2-Fluoroamphetamine

1 gram of 2-FA
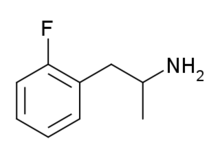
2-Fluoroamphetamine (2-FA) is a stimulant drug from the amphetamine family which has been sold as a designer drug.[1] 2-Fluoroamphetamine differs from 3- and 4-fluoroamphetamine in the position of the fluorine atom on the aromatic ring, making them positional isomers of one another. The replacement of a hydrogen atom with a fluorine atom in certain compounds to facilitate passage through the blood–brain barrier, as is desirable in central nervous system pharmaceutical agents, is a common practice due to the corresponding increase in lipophilicity granted by the substitute.[2][3]
Pharmacology
Anorexiant dose (amount inhibiting food intake by 50% for 2 hours, given 1 hour earlier) = 15 mg/kg (rat; p.o.).[4]
Analgesic dose (50% inhibition of response to tail-clamp) = 20 mg/kg (mouse; i.p.).[4]
Effect on blood pressure: 0.5 mg/kg (rat; i.v.) produces an increase in BP of 29 mm.[4]
Toxicology
LD50 (mouse; i.p.) = 100 mg/kg.[4]
Legal Status
United States
The Federal Analog Act, 21 U.S.C. § 813, is a section of the United States Controlled Substances Act, allowing any chemical "substantially similar" to an illegal drug (in Schedule I or II) to be treated as if it were also in Schedule I or II, but only if it is intended (ref 1) for human consumption. 2-FA may be considered to be an analog of amphetamine, thus falling under the Federal Analog Act.
China
As of October 2015 2-FA is a controlled substance in China.[5]
See also
References
- ↑ Rösner, P; Quednow, B; Girreser, U; Junge, T (2005). "Isomeric fluoro-methoxy-phenylalkylamines: a new series of controlled-substance analogues (designer drugs)". Forensic Science International. 148 (2–3): 143–56. doi:10.1016/j.forsciint.2004.05.003. PMID 15639609.
- ↑ B.E. Smart (2001). "Fluorine substituent effects (on bioactivity". Journal of Fluorine Chemistry. 109: 3–11. doi:10.1016/s0022-1139(01)00375-x.
- ↑ F.M.D. Ismail (2002). "Important fluorinated drugs in experimental and clinical use". Journal of Fluorine Chemistry. 118: 27–33. doi:10.1016/s0022-1139(02)00201-4.
- 1 2 3 4 E. Costa and S. Garattini (1970) Amphetamines and Related Compounds, p.28, New York: Raven Press.
- ↑ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. 27 September 2015. Retrieved 1 October 2015.
|
|---|
|
| Adamantanes | |
|---|
|
| Adenosine antagonists | |
|---|
|
| Alkylamines | |
|---|
|
| Ampakines | |
|---|
|
| Arylcyclohexylamines | |
|---|
|
| Benzazepines | |
|---|
|
| Cholinergics | |
|---|
|
| Convulsants | |
|---|
|
| Eugeroics | |
|---|
|
| Oxazolines | |
|---|
|
| Phenethylamines |
- 1-(4-Methylphenyl)-2-aminobutane
- 1-Phenyl-2-(piperidin-1-yl)pentan-3-one
- 1-Methylamino-1-(3,4-methylenedioxyphenyl)propane
- 2-Fuoroamphetamine
- 2-Fuoromethamphetamine
- 2-OH-PEA
- 2-Phenyl-3-aminobutane
- 2-Phenyl-3-methylaminobutane
- 2,3-MDA
- 3-Fuoroamphetamine
- 3-Fluoroethamphetamine
- 3-Fluoromethcathinone
- 3-Methoxyamphetamine
- 3-Methylamphetamine
- 3,4-DMMC
- 4-BMC
- 4-CMC
- 4-Ethylamphetamine
- 4-Fluoroamphetamine
- 4-Fluoromethamphetamine
- 4-MA
- 4-Methylbuphedrone
- 4-Methylcathinone
- 4-MMA
- 4-Methylpentedrone
- 4-MTA
- 6-FNE
- AL-1095
- Alfetamine
- a-Ethylphenethylamine
- Amfecloral
- Amfepentorex
- Amfepramone
- Amidephrine
- 2-Amino-1,2-dihydronaphthalene
- 2-Aminoindane
- 5-(2-Aminopropyl)indole
- 2-Aminotetralin
- Acridorex
- Amphetamine (Dextroamphetamine, Levoamphetamine)
- Amphetaminil
- Arbutamine
- β-Methylphenethylamine
- β-Phenylmethamphetamine
- Benfluorex
- Benzedrone
- Benzphetamine
- BDB
- BOH
- 3-Benzhydrylmorpholine
- BPAP
- Buphedrone
- Bupropion
- Butylone
- Camfetamine
- Cathine
- Cathinone
- Chlorphentermine
- Cilobamine
- Cinnamedrine
- Clenbuterol
- Clobenzorex
- Cloforex
- Clortermine
- Cypenamine
- D-Deprenyl
- Denopamine
- Dimethoxyamphetamine
- Dimethylamphetamine
- Dimethylcathinone
- Dobutamine
- DOPA (Dextrodopa, Levodopa)
- Dopamine
- Dopexamine
- Droxidopa
- EBDB
- Ephedrine
- Epinephrine
- Epinine
- Etafedrine
- Ethcathinone
- Ethylnorepinephrine
- Ethylone
- Etilamfetamine
- Etilefrine
- Famprofazone
- Fencamfamine
- Fencamine
- Fenethylline
- Fenfluramine (Dexfenfluramine, Levofenfluramine)
- Fenproporex
- Feprosidnine
- Flephedrone
- Fludorex
- Formetorex
- Furfenorex
- Gepefrine
- Hexapradol
- Hexedrone
- HMMA
- Hordenine
- 4-Hydroxyamphetamine
- 5-Iodo-2-aminoindane
- Ibopamine
- IMP
- Indanylamphetamine
- Iofetamine
- Isoetarine
- Isoethcathinone
- Isoprenaline
- L-Deprenyl (Selegiline)
- Lefetamine
- Lisdexamfetamine
- Lophophine
- MBDB
- MDA
- MDBU
- MDEA
- MDMA
- MDMPEA
- MDOH
- MDPR
- MDPEA
- Mefenorex
- Mephedrone
- Mephentermine
- Metanephrine
- Metaraminol
- Mesocarb
- Methamphetamine (Dextromethamphetamine, Levomethamphetamine)
- Methoxamine
- Methoxyphenamine
- MMA
- Methcathinone
- Methedrone
- Methoxyphenamine
- Methylenedioxycathinone
- Methylone
- Mexedrone
- MMDA
- MMDMA
- MMMA
- Morforex
- N,alpha-Diethylphenylethylamine
- N-Benzyl-1-phenethylamine
- N-Ethylbuphedrone
- N-Ethylhexedrone
- N,N-Dimethylphenethylamine
- Naphthylamphetamine
- Nisoxetine
- Norepinephrine
- Norfenefrine
- Norfenfluramine
- Normetanephrine
- L-Norpseudoephedrine
- Octopamine (drug)
- Orciprenaline
- Ortetamine
- Oxifentorex
- Oxilofrine
- PBA
- PCA
- PCMA
- PHA
- Pentorex
- Pentedrone
- Pentylone
- Phenatine
- Phenpromethamine
- Phentermine
- Phenylalanine
- Phenylephrine
- Phenylpropanolamine
- Pholedrine
- PIA
- PMA
- PMEA
- PMMA
- PPAP
- Phthalimidopropiophenone
- Prenylamine
- Propylamphetamine
- Pseudoephedrine
- Ropinirole
- Salbutamol (Levosalbutamol)
- Sibutramine
- Synephrine
- Theodrenaline
- Tiflorex
- Tranylcypromine
- Tyramine
- Tyrosine
- Xylopropamine
- Zylofuramine
|
|---|
|
| Phenylmorpholines | |
|---|
|
| Piperazines | |
|---|
|
| Piperidines | |
|---|
|
| Pyrrolidines | |
|---|
|
| Racetams | |
|---|
|
| Tropanes | |
|---|
|
| Tryptamines | |
|---|
|
| Others | |
|---|
|
|