Cumene
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
(Propan-2-yl)benzene | |||
| Other names | |||
| Identifiers | |||
| 98-82-8 | |||
| 3D model (Jmol) | Interactive image | ||
| ChEBI | CHEBI:34656 | ||
| ChemSpider | 7128 | ||
| ECHA InfoCard | 100.002.458 | ||
| KEGG | C14396 | ||
| PubChem | 7406 | ||
| RTECS number | GR8575000 | ||
| UNII | 8Q54S3XE7K | ||
| |||
| |||
| Properties | |||
| C9H12 | |||
| Molar mass | 120.20 g·mol−1 | ||
| Appearance | colorless liquid | ||
| Odor | sharp, gasoline-like | ||
| Density | 0.862 g cm−3, liquid | ||
| Melting point | −96 °C (−141 °F; 177 K) | ||
| Boiling point | 152 °C (306 °F; 425 K) | ||
| negligible | |||
| Solubility | soluble in acetone, ether, ethanol | ||
| Vapor pressure | 8 mm (20°C)[2] | ||
| Refractive index (nD) |
1.4915 (20 °C) | ||
| Viscosity | 0.777 cP (21 °C) | ||
| Hazards | |||
| Main hazards | flammable | ||
| R-phrases | R10,R37,R51/53,R65 | ||
| S-phrases | S24,S37,S61,S62 | ||
| NFPA 704 | |||
| Flash point | 43 °C (109 °F; 316 K) | ||
| 424 °C (795 °F; 697 K) | |||
| Explosive limits | 0.9-6.5% | ||
| Lethal dose or concentration (LD, LC): | |||
| LD50 (median dose) |
12750 mg/kg (oral, mouse) 1400 mg/kg (oral, rat)[3] | ||
| LC50 (median concentration) |
200 ppm (mouse, 7 hr)[3] | ||
| LCLo (lowest published) |
8000 ppm (rat, 4 hr)[3] | ||
| US health exposure limits (NIOSH): | |||
| PEL (Permissible) |
TWA 50 ppm (245 mg/m3) [skin][2] | ||
| REL (Recommended) |
TWA 50 ppm (245 mg/m3) [skin][2] | ||
| IDLH (Immediate danger) |
900 ppm[2] | ||
| Related compounds | |||
| Related compounds |
ethylbenzene toluene benzene | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Cumene is the common name for isopropylbenzene, an organic compound that is based on an aromatic hydrocarbon with an aliphatic substitution. It is a constituent of crude oil and refined fuels. It is a flammable colorless liquid that has a boiling point of 152 °C. Nearly all the cumene that is produced as a pure compound on an industrial scale is converted to cumene hydroperoxide, which is an intermediate in the synthesis of other industrially important chemicals, primarily phenol and acetone.
Production
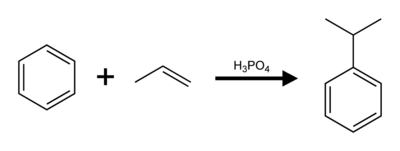
Commercial production of cumene is by Friedel–Crafts alkylation of benzene with propylene. Cumene producers account for approximately 20% of the global demand for benzene.[4] Previously, solid phosphoric acid (SPA) supported on alumina was used as the catalyst. Since the mid-1990s, commercial production has switched to zeolite-based catalysts.[5]
Isopropylbenzene is stable, but may form peroxides in storage if in contact with the air. It is important to test for the presence of peroxides before heating or distilling. The chemical is also flammable and incompatible with strong oxidizing agents.
Health effects
A review by the US Department of Health and Human Services found that mice exposed to cumene fumes developed tumors in their lungs and livers. Cumene was anticipated to also be a human carcinogen and was added to the governmental organization's list of carcinogens in 2014.[6]
See also
References
- ↑ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. pp. 139, 597. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- 1 2 3 4 "NIOSH Pocket Guide to Chemical Hazards #0159". National Institute for Occupational Safety and Health (NIOSH).
- 1 2 3 "Cumene". Immediately Dangerous to Life and Health. National Institute for Occupational Safety and Health (NIOSH).
- ↑ Market Study Benzene, published by Ceresana, July 2011
- ↑ The Innovation Group website, page accessed 15/11/07
- ↑ Gannon, Megan (October 3, 2014). "4 New Substances Added To List Of Carcinogens". Huffington Post. LiveScience.
External links
- National Pollutant Inventory - Cumene fact sheet
- CDC - NIOSH Pocket Guide to Chemical Hazards
- Cumene Production from Benzene and Propylene Using Aluminum Chloride Catalyst