Bicarbonate
 | |
 | |
| Names | |
|---|---|
| Systematic IUPAC name
Hydroxidodioxidocarbonate(1−)[1] | |
| Other names
Hydrogencarbonate[1] | |
| Identifiers | |
| 71-52-3 | |
| 3D model (Jmol) | Interactive image |
| 3DMet | B00080 |
| 3903504 | |
| ChEBI | CHEBI:17544 |
| ChEMBL | ChEMBL363707 |
| ChemSpider | 749 |
| 49249 | |
| KEGG | C00288 |
| PubChem | 769 |
| UNII | HN1ZRA3Q20 |
| |
| |
| Properties | |
| HCO− 3 | |
| Molar mass | 61.0168 g mol−1 |
| log P | −0.82 |
| Acidity (pKa) | 10.3 (Conjugate acid of carbonate) |
| Basicity (pKb) | 7.7 (Conjugate base of carbonic acid) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
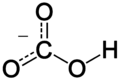
In inorganic chemistry, bicarbonate (IUPAC-recommended nomenclature: hydrogen carbonate[2]) is an intermediate form in the deprotonation of carbonic acid. It is a polyatomic anion with the chemical formula HCO−
3.
Bicarbonate serves a crucial biochemical role in the physiological pH buffering system.[3]
The term "bicarbonate" was coined in 1814 by the English chemist William Hyde Wollaston.[4] The prefix "bi" in "bicarbonate" comes from an outdated naming system and is based on the observation that there is twice as much carbonate (CO2−
3) per sodium ion in sodium bicarbonate (NaHCO3) and other bicarbonates than in sodium carbonate (Na2CO3) and other carbonates.[5] The name lives on as a trivial name.
Chemical properties
The bicarbonate ion (hydrogencarbonate ion) is an anion with the empirical formula HCO−
3 and a molecular mass of 61.01 daltons; it consists of one central carbon atom surrounded by three oxygen atoms in a trigonal planar arrangement, with a hydrogen atom attached to one of the oxygens. It is isoelectronic with nitric acid HNO
3. The bicarbonate ion carries a negative one formal charge and is the conjugate base of carbonic acid H
2CO
3; at the same time, it is the conjugate acid of CO2−
3, the carbonate ion, as shown by these equilibrium reactions:
- CO2−
3 + 2 H2O ⇌ HCO−
3 + H2O + OH− ⇌ H2CO3 + 2 OH−
- H2CO3 + 2 H2O ⇌ HCO−
3 + H3O+ + H2O ⇌ CO2−
3 + 2 H3O+.
A bicarbonate salt forms when a positively charged ion attaches to the negatively charged oxygen atoms of the ion, forming an ionic compound. Many bicarbonates are soluble in water at standard temperature and pressure; in particular, sodium bicarbonate contributes to total dissolved solids, a common parameter for assessing water quality.
Physiological role
Bicarbonate (HCO−
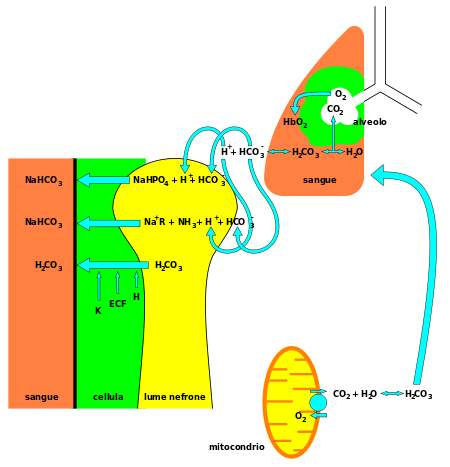
3) is alkaline, and a vital component of the pH buffering system[3] of the human body (maintaining acid-base homeostasis). 70–75% of CO2 in the body is converted into carbonic acid (H2CO3), which can quickly turn into bicarbonate.
With carbonic acid as the central intermediate species, bicarbonate – in conjunction with water, hydrogen ions, and carbon dioxide – forms this buffering system, which is maintained at the volatile equilibrium[3] required to provide prompt resistance to pH changes in both the acidic and basic directions. This is especially important for protecting tissues of the central nervous system, where pH changes too far outside of the normal range in either direction could prove disastrous (see acidosis or alkalosis).
Bicarbonate also acts to regulate pH in the small intestine. It is released from the pancreas in response to the hormone secretin to neutralize the acidic chyme entering the duodenum from the stomach.[6]
Bicarbonate in the environment
In freshwater ecology, strong photosynthetic activity by freshwater plants in daylight releases gaseous oxygen into the water and at the same time produces bicarbonate ions. These shift the pH upward until in certain circumstances the degree of alkalinity can become toxic to some organisms or can make other chemical constituents such as ammonia toxic. In darkness, when no photosynthesis occurs, respiration processes release carbon dioxide, and no new bicarbonate ions are produced, resulting in a rapid fall in pH.
Other uses
The most common salt of the bicarbonate ion is sodium bicarbonate, NaHCO3, which is commonly known as baking soda. When heated or exposed to an acid such as acetic acid (vinegar), sodium bicarbonate releases carbon dioxide. This is used as a leavening agent in baking.
The flow of bicarbonate ions from rocks weathered by the carbonic acid in rainwater is an important part of the carbon cycle.
Bicarbonate also serves much in the digestive system. It raises the internal pH of the stomach, after highly acidic digestive juices have finished in their digestion of food. Ammonium bicarbonate is used in digestive biscuit manufacture.
Diagnostics
In diagnostic medicine, the blood value of bicarbonate is one of several indicators of the state of acid–base physiology in the body. It is measured, along with carbon dioxide, chloride, potassium, and sodium, to assess electrolyte levels in an electrolyte panel test (which has Current Procedural Terminology, CPT, code 80051).
The parameter standard bicarbonate concentration (SBCe) is the bicarbonate concentration in the blood at a PaCO2 of 40 mmHg (5.33 kPa), full oxygen saturation and 36 °C.[7]
Bicarbonate compounds
- Sodium bicarbonate
- Potassium bicarbonate
- Caesium bicarbonate
- Magnesium bicarbonate
- Calcium bicarbonate
- Ammonium bicarbonate
- Carbonic acid
See also
References
- 1 2 "hydrogencarbonate (CHEBI:17544)". Chemical Entities of Biological Interest (ChEBI). UK: European Institute of Bioinformatics. IUPAC Names.
- ↑ Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005 (PDF), IUPAC, p. 137
- 1 2 3 "Clinical correlates of pH levels: bicarbonate as a buffer". Biology.arizona.edu. October 2006.
- ↑ William Hyde Wollaston (1814) "A synoptic scale of chemical equivalents," Philosophical Transactions of the Royal Society, 104: 1-22. On page 11, Wollaston coins the term "bicarbonate": "The next question that occurs relates to the composition of this crystallized carbonate of potash, which I am induced to call bi-carbonate of potash, for the purpose of marking more decidedly the distinction between this salt and that which is commonly called a subcarbonate, and in order to refer at once to the double dose of carbonic acid contained in it."
- ↑ http://www.newton.dep.anl.gov/askasci/chem99/chem99492.htm
- ↑ Berne & Levy, Principles of Physiology
- ↑ Acid Base Balance (page 3)
External links
| Look up bicarbonate in Wiktionary, the free dictionary. |
- Bicarbonates at the US National Library of Medicine Medical Subject Headings (MeSH)