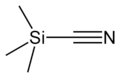Trimethylsilyl cyanide
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
trimethylsilylformonitrile | |||
| Other names
Cyanotrimethylsilane; TMS cyanide; Trimethylsilylnitrile; Trimethylsilanecarbonitrile' Trimethylsilylcarbonitrile | |||
| Identifiers | |||
| 7677-24-9 | |||
| 3D model (Jmol) | Interactive image | ||
| Abbreviations | TMSCN | ||
| ChemSpider | 74110 | ||
| ECHA InfoCard | 100.028.780 | ||
| PubChem | 82115 | ||
| |||
| |||
| Properties | |||
| C4H9NSi | |||
| Molar mass | 99.21 g·mol−1 | ||
| Density | 0.793 g/mL at 20 °C | ||
| Melting point | 8 to 11 °C (46 to 52 °F; 281 to 284 K) | ||
| Boiling point | 114 to 117 °C (237 to 243 °F; 387 to 390 K) | ||
| reacts with water | |||
| Solubility | organic solvents | ||
| Refractive index (nD) |
1.392 | ||
| Hazards | |||
| R-phrases | R11 R26/27/28 R29 | ||
| S-phrases | S16 S36/37/39 S45 | ||
| Flash point | 1 °C (34 °F; 274 K) | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Trimethylsilyl cyanide is the chemical compound with the formula (CH3)3SiCN. This volatile liquid consists of a cyanide group, that is CN, attached to a trimethylsilyl group. The molecule is used in organic synthesis as the equivalent of hydrogen cyanide. It is prepared by the reaction of lithium cyanide and trimethylsilyl chloride:[1]
- LiCN + (CH3)3SiCl → (CH3)3SiCN + LiCl
Structure
The molecule exhibits the expected structure of a nitrile-like compound. The compound exists in a facile equilibrium with a small amount of the isomeric isocyanide (CH3)3SiNC.[2] By contrast, the nearly isostructural tert-butylnitrile does not readily isomerize to tert-butyl isocyanide.
Reactions
Trimethylsilyl cyanide hydrolyzes to give hydrogen cyanide:
- (CH3)3SiCN + H2O → (CH3)3SiOH + HCN
In its principal application, it adds across carbon-oxygen double bonds, for example in an aldehyde:
- RCHO + (CH3)3SiCN → RCH(CN)OSi(CH3)3
The product is an O-silylated cyanohydrin.
One use of this reagent is to convert pyridine-N-oxides into 2-cyanopyridine. This transformation is best done in dichloromethane solution using dimethylcarbamoyl chloride as the activating electrophile. It is possible to use benzoyl chloride but the yields and regioselectivity of the addition of the cyano group are lower.
Safety
This chemical is hazardous as it hydrolyzes in moist air to give hydrogen cyanide.
References
- ↑ Livinghouse, T. (1990). "Trimethylsilyl Cyanide: Cyanosilation of p-Benzoquinone". Org. Synth.; Coll. Vol., 7, p. 517
- ↑ M. R. Booth and S. G. Frankiss "Trimethylsilyl isocyanide" Chem. Commun. 1968, pp. 1347-1348. doi:10.1039/C19680001347