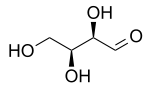
Threose
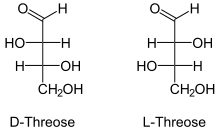
 D-Threose | |
 L-Threose | |
| Names | |
|---|---|
| IUPAC names
(2S,3R)-2,3,4-Trihydroxybutanal (D) (2R,3S)-2,3,4-Trihydroxybutanal (L) | |
| Other names
Threotetrose | |
| Identifiers | |
| 95-43-2 (D) 95-44-3 (L) | |
| 3D model (Jmol) | (D): Interactive image (L): Interactive image |
| ChEBI | CHEBI:28587 |
| ChemSpider | 388736 |
| ECHA InfoCard | 100.002.199 |
| PubChem | 439665 (D) |
| |
| |
| Properties | |
| C4H8O4 | |
| Molar mass | 120.10 g·mol−1 |
| Appearance | Syrup |
| Very soluble | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Threose is a four-carbon monosaccharide or carbohydrate with molecular formula C4H8O4. It has a terminal aldehyde group rather than a ketone in its linear chain, and so is considered part of the aldose family of monosaccharides. The threose name can be used to refer to both the D- and L-stereoisomers, and more generally to the racemic mixture (D/L-, equal parts D- and L-) as well as to the more generic threose structure (absolute stereochemistry unspecified).
The prefix "threo" which derives from threose (and "erythro" from a corresponding diastereomer erythrose) offer a useful way to describe general organic structures with adjacent chiral centers, where "the prefixes... designate the relative configuration of the centers".[2] As is depicted in a Fischer projection of D-threose, the adjacent substituents will have a syn orientation in the isomer referred to as "threo", and are anti in the isomer referred to as "erythro".[2][3]
See also
References
- ↑ Merck Index, 11th Edition, 9317
- 1 2 Formulas Using Other Configurational Notations, W. Rausch, accessed 1 March 2011
- ↑ Prof. Rausch helpfully notes that the prefixes "may be applied to racemic compounds, as well as pure enantiomers and meso compounds", and that when depicted in the common "zig-zag" representation, adjacent "substituents may lie on the same side of the carbon chain... [syn] or on opposite sides... [anti]", which is opposite of their depiction in a Fischer projection.