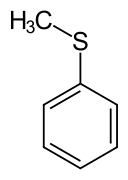
Thioanisole
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(Methylsulfanyl)benzene | |
| Other names | |
| Identifiers | |
| 100-68-5 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 7239 |
| EC Number | 202-878-2 |
| PubChem | 7520 |
| |
| |
| Properties | |
| C7H8S | |
| Molar mass | 124.20 |
| Appearance | colorless liquid |
| Density | 1.0533 g/cm3 |
| Melting point | -15 ºC |
| Boiling point | 193 ºC |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Thioanisole is an organic compound with the formula CH3SC6H5. It is a colorless liquid, which is soluble in organic solvents. It is the simplest alkyl-aryl thioether. The name indicates that this compound is the sulfur-analogue of methoxybenzene, anisole.
It can be prepared by methylation of thiophenol.
Reactions
Thioanisole undergoes deprotonation with alkyllithium reagents to afford C6H5SCH2Li, which can be alkylated. The resulting homologated thioether can be manipulated in a variety of ways.[2]
Oxidation of thioanisole gives the chiral sulfoxide.[3]
References
- ↑ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 707. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ↑ Bailey, Simon “Thioanisole” e-EROS Encyclopedia of Reagents for Organic Synthesis, 2001, John Wiley & Sons. doi:10.1002/047084289X.rt093
- ↑ Carl R. Johnson, Jeffrey E. Keiser "Methyl Phenyl Sulfoxide" Org. Synth. 1966, volume 46, 78. doi:10.15227/orgsyn.046.0078
This article is issued from Wikipedia - version of the 9/24/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.