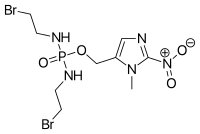
Evofosfamide
 | |
| Names | |
|---|---|
| IUPAC name
(1-Methyl-2-nitro-1H-imidazol-5-yl)methyl N,N'-bis(2-bromoethyl)phosphorodiamidate | |
| Other names
TH-302; HAP-302 | |
| Identifiers | |
| 918633-87-1 | |
| 3D model (Jmol) | Interactive image |
| ChEMBL | ChEMBL260046 |
| ChemSpider | 10157061 |
| PubChem | 11984561 |
| |
| |
| Properties | |
| C9H16Br2N5O4P | |
| Molar mass | 449.04 g·mol−1 |
| 6 to 7 g/l | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Evofosfamide (INN,[1] USAN;[2] formerly known as TH-302) is an investigational hypoxia-activated prodrug that is in clinical development for cancer treatment. The prodrug is activated only at very low levels of oxygen (hypoxia). Such levels are common in human solid tumors, a phenomenon known as tumor hypoxia.[3]
Evofosfamide is being evaluated in clinical trials for the treatment of multiple tumor types as a monotherapy and in combination with chemotherapeutic agents and other targeted cancer drugs.
Dec 2015 : two Phase 3 trials fail, Merck will not apply for a license
Collaboration
Evofosfamide was developed by Threshold Pharmaceuticals Inc. In 2012, Threshold signed a global license and co-development agreement for evofosfamide with Merck KGaA, Darmstadt, Germany (EMD Serono Inc. in the US and Canada), which includes an option for Threshold to co-commercialize evofosfamide in the United States. Threshold is responsible for the development of evofosfamide in the soft tissue sarcoma indication in the United States. In all other cancer indications, Threshold and Merck KGaA are developing evofosfamide together.[4] From 2012 to 2013, Merck KGaA paid 110 million US$ for upfront payment and milestone payments to Threshold. Additionally, Merck KGaA covers 70% of all evofosfamide development expenses.[5]
Mechanism of prodrug activation and Mechanism of action (MOA) of the released drug
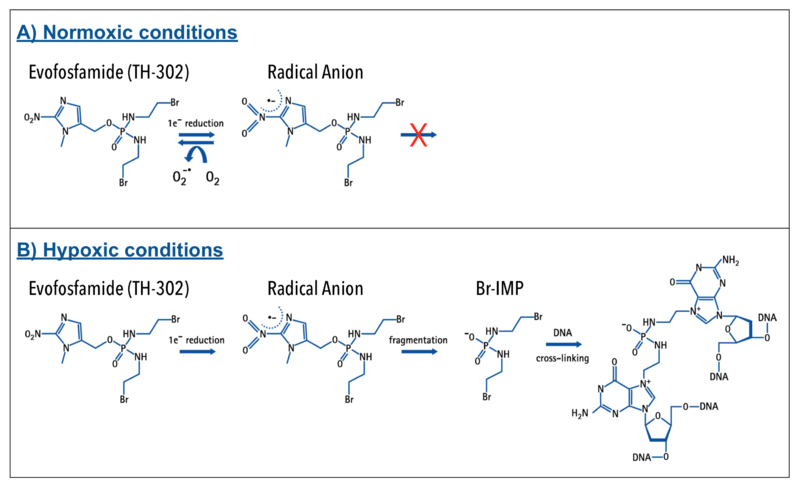
Evofosfamide is a 2-nitroimidazole prodrug of the cytotoxin bromo-isophosphoramide mustard (Br-IPM). Evofosfamide is activated by a process that involves a 1-electron (1 e−) reduction mediated by ubiquitous cellular reductases, such as the NADPH cytochrome P450, to generate a radical anion prodrug:
- A) In the presence of oxygen (normoxia) the radical anion prodrug reacts rapidly with oxygen to generate the original prodrug and superoxide. Therefore, evofosfamide is relatively inert under normal oxygen conditions, remaining intact as a prodrug.
- B) When exposed to severe hypoxic conditions (< 0.5% O2; hypoxic zones in many tumors), however, the radical anion undergoes irreversible fragmentation, releasing the active drug Br-IPM and an azole derivative. The released cytotoxin Br-IPM alkylates DNA, inducing intrastrand and interstrand crosslinks.[6]
Evofosfamide is essentially inactive under normal oxygen levels. In areas of hypoxia, evofosfamide becomes activated and converts to an alkylating cytotoxic agent resulting in DNA cross-linking. This renders cells unable to replicable their DNA and divide, leading to apoptosis. This investigational therapeutic approach of targeting the cytotoxin to hypoxic zones in tumors may cause less broad systemic toxicity that is seen with untargeted cytotoxic chemotherapies.[7]
The activation of evofosfamide to the active drug Br-IPM and the mechanism of action (MOA) via cross-linking of DNA is shown schematically below:
Drug development history
Phosphorodiamidate-based, DNA-crosslinking, bis-alkylator mustards have long been used successfully in cancer chemotherapy and include e.g. the prodrugs ifosfamide and cyclophosphamide. To demonstrate that known drugs of proven efficacy could serve as the basis of efficacious hypoxia-activated prodrugs, the 2-nitroimidizole HAP of the active phosphoramidate bis-alkylator derived from ifosfamide was synthesized. The resulting compound, TH-281, had a high HCR (hypoxia cytotoxicity ratio), a quantitative assessment of its hypoxia selectivity. Subsequent structure-activity relationship (SAR) studies showed that replacement of the chlorines in the alkylator portion of the prodrug with bromines improved potency about 10-fold. The resulting, final compound is evofosfamide (TH-302).[8]
Synthesis
Evofosfamide can be synthesized in 7 steps.[9][10]
Formulation
The evofosfamide drug product formulation used until 2011 was a lyophilized powder. The current drug product formulation is a sterile liquid containing ethanol, dimethylacetamide and polysorbate 80. For intravenous infusion, the evofosfamide drug product is diluted in 5% dextrose in WFI.[11]
Diluted evofosfamide formulation (100 mg/ml evofosfamide, 70% ethanol, 25% dimethylacetamide and 5% polysorbate 80; diluted to 4% v/v in 5% dextrose or 0.9% NaCl) can cause leaching of DEHP from infusion bags containing PVC plastic.[12]
Clinical trials
Overview and results
Evofosfamide (TH-302) is currently being evaluated in clinical studies as a monotherapy and in combination with chemotherapy agents and other targeted cancer drugs. The indications are a broad spectrum of solid tumor types and blood cancers.
Evofosfamide clinical trials (as of 21 November 2014)[13] sorted by (Estimated) Primary Completion Date:[14]
| Evofosfamide (TH-302) clinical trials in following indications:
Solid Tumors, Soft Tissue Sarcoma, Pancreatic Cancer/Pancreatic Adenocarcinoma/Pancreatic Neuroendocrine Tumors, Non-Small Cell Lung Cancer, Melanoma, Prostate Cancer, Kidney Cancer/Renal Cell Carcinoma, Liver Cancer/Hepatocellular Carcinoma, Myelogenous Leukemia/Lymphocytic Leukemia, Myelodysplastic Syndrome, Myelofibrosis, Multiple Myeloma, Glioma/Glioblastoma, Gastrointestinal Stromal Tumors | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| NCT Number / Title | Other IDs | Start Date | (Estimated) Primary Completion Date | Recruitment | Conditions | Interventions | Phases | Enrollment (Patients) | Sponsor / Collaborators |
| NCT00495144 A Phase I Study of the Safety, Pharmacokinetics, and Anti-Tumor Activity of TH-302 in Patients With Advanced Solid Tumors | TH-CR-401 | June 2007 | December 2011 | Completed | Tumors, Hypoxia | Drug: TH-302 | Phase 1 | 129 | Threshold Pharmaceuticals |
| NCT01149915 Study of Hypoxia-Activated Prodrug TH-302 to Treat Advanced Leukemias | TH-CR-407 | June 2010 | August 2013 | Completed | Acute Myelogenous Leukemia, Acute Lymphoblastic Leukemia, Chronic Myelogenous Leukemia, High-risk Myelodysplastic Syndrome, Chronic Lymphocytic Leukemia, Advanced Myelofibrosis | Drug: TH-302 | Phase 1 | 40 | Threshold Pharmaceuticals |
| NCT01144455 Study of the Safety and Efficacy of TH-302 in Combination With Gemcitabine Compared With Gemcitabine Alone in Previously Untreated Patients With Pancreatic Adenocarcinoma |
TH-CR-404 | June 2010 | August 2013 | Completed | Pancreatic Adenocarcinoma | Drug: Gemzar (Gemcitabine), Drug: TH-302, Drug: TH-302 | Phase 2 | 165 | Threshold Pharmaceuticals, PRA Health Sciences |
Results:
18 November 2014[17]
| |||||||||
| NCT00742963 Dose-Escalation Study of TH-302 in Combination With Doxorubicin to Treat Patients With Advanced Soft Tissue Sarcoma | TH-CR-403 | August 2008 | September 2013 | Completed | Soft Tissue Sarcoma | Drug: TH-302 | Phase 1, Phase 2 | 102 | Threshold Pharmaceuticals |
| Results:
15 November 2012, Results for overall study population (N=91)[18][19]
10 October 2014, Results for overall study population (N=91)[20]
| |||||||||
| NCT00743379 Dose-Escalation Study of TH-302 in Combination With A) Gemcitabine or B) Docetaxel or C) Pemetrexed to Treat Advanced Solid Tumors | TH-CR-402 | August 2008 | September 2013 | Completed | Non-Small Cell Lung Cancer, Prostate Cancer, Pancreatic Cancer | Drug: TH-302 | Phase 1, Phase 2 | 71 | Threshold Pharmaceuticals |
| Results:
12 October 2010[21]
| |||||||||
| NCT01497444 Sorafenib Tosylate and Hypoxia-Activated Prodrug TH-302 in Treating Patients With Advanced Kidney Cancer or Liver Cancer That Cannot Be Removed By Surgery | TH-IST-4004, N1153, NCCTG-N1153, CDR0000720022, NCI-2012-00095, U10CA031946 | May 2012 | January 2014 | Recruiting | Kidney Cancer, Liver Cancer | Drug: hypoxia-activated prodrug TH-302, Drug: sorafenib tosylate, Genetic: protein analysis, Other: laboratory biomarker analysis | Phase 1, Phase 2 | 48 | North Central Cancer Treatment Group, National Cancer Institute (NCI), Alliance for Clinical Trials in Oncology, Cancer and Leukemia Group B, American College of Surgeons, Threshold Pharmaceuticals |
| NCT01440088 A Trial of TH-302 in Combination With Doxorubicin Versus Doxorubicin Alone to Treat Patients With Locally Advanced Unresectable or Metastatic Soft Tissue Sarcoma | TH-CR-406 / SARC021 | September 2011 | June 2014 | Active, not recruiting | Soft Tissue Sarcoma | Drug: TH-302 in Combination with Doxorubicin, Drug: Doxorubicin | Phase 3 | 620 | Threshold Pharmaceuticals, Sarcoma Alliance for Research through Collaboration (SARC) |
| Results:
18 September 2014[26]
22 September 2014[27]
3 November 2014[28]
| |||||||||
| NCT01381822 Dose-Escalation Study of TH-302 in Combination With Sunitinib to Treat Patients With Advanced Renal Cell Carcinoma,Gastrointestinal Stromal Tumors and Pancreatic Neuroendocrine Tumors | TH-CR-410 | June 2011 | June 2014 | Active, not recruiting | Advanced Renal Cell Carcinoma, Gastrointestinal Stromal Tumors, Pancreatic Neuroendocrine Tumors | Drug: TH-302 | Phase 1 | 58 | Threshold Pharmaceuticals |
| NCT01833546 A Japanese Phase 1 Trial of TH-302 in Subjects With Solid Tumors and Pancreatic Cancer | EMR200592-002 | April 2013 | January 2015 | Active, not recruiting | Solid Tumor, Pancreatic Cancer | Drug: TH-302 monotherapy, Drug: TH-302, Drug: Gemcitabine | Phase 1 | 20 | Merck KGaA, Threshold Pharmaceuticals |
| NCT02020226 A Cardiac Safety Study of TH-302 in Patients With Advanced Solid Tumors | TH-CR-414 | November 2013 | January 2015 | Recruiting | Solid Tumors | Drug: TH-302 | Phase 1 | 40 | Threshold Pharmaceuticals |
| NCT02076230 A Phase 1 TH-302 Mass Balance Trial | EMR200592-007, 2013-003950-25 | February 2014 | March 2015 | Active, not recruiting | Solid Tumors | Drug: [14C] TH-302 (Label 1), Drug: [14C] TH-302 (Label 2), Drug: Unlabeled TH-302 | Phase 1 | 10 | Merck KGaA, Threshold Pharmaceuticals |
| NCT01522872 Open-label Study of TH-302 and Dexamethasone With or Without Bortezomib in Subjects With Relapsed/Refractory Multiple Myeloma | TH-CR-408 | February 2012 | June 2015 | Recruiting | Multiple Myeloma | Drug: TH-302, Drug: TH-302, Drug: TH-302 and bortezomib | Phase 1, Phase 2 | 60 | Threshold Pharmaceuticals |
| NCT01864538 A Phase 2 Biomarker - Enriched Study of TH-302 in Subjects With Advanced Melanoma | TH-CR-413 | May 2013 | June 2015 | Recruiting | Metastatic Melanoma | Drug: TH-302 | Phase 2 | 40 | Threshold Pharmaceuticals |
| NCT01403610 Safety and Efficacy Study of TH-302 CNS Penetration in Recurrent High Grade Astrocytoma Following Bevacizumab | TH-IST-4003, CTRC 11-24 | June 2011 | July 2015 | Recruiting | High Grade Glioma | Drug: TH-302, Drug: Placebo | Phase 2 | 34 | The University of Texas Health Science Center at San Antonio |
Results:
17 November 2014[31]
| |||||||||
| NCT01721941 TH-302 Plus Doxorubicin Delivered by Trans-Arterial Chemoembolization (TACE) in Patients With Hepatocellular Carcinoma | TH302 TACE | December 2014 | December 2015 | Not yet recruiting | Hepatocellular Carcinoma | Drug: Phase I Dose level -1, Drug: Phase I dose level 1, Drug: Phase I Dose level 2, Drug: Phase I Dose level 3 | Phase 1 | 20 | Scripps Clinic Cancer Center, Threshold Pharmaceuticals |
| NCT01746979 Clinical Trial Testing TH-302 in Combination With Gemcitabine in Previously Untreated Subjects With Metastatic or Locally Advanced Unresectable Pancreatic Adenocarcinoma | MAESTRO, EMR 200592-001, 2012-002957-42 | December 2012 | February 2016 | Active, not recruiting | Metastatic or Locally Advanced Unresectable Pancreatic Adenocarcinoma | Drug: TH-302, Drug: Gemcitabine, Drug: Placebo (5 percent dextrose - D5W) | Phase 3 | 660 | EMD Serono, Threshold Pharmaceuticals |
| Results:
3 November 2014[32]
18 November 2014[33]
| |||||||||
| NCT02047500 Phase I TH-302 Plus Gemcitabine Plus Nab-Paclitaxel in Pancreatic Cancer | EMR200592-006 | January 2014 | June 2016 | Recruiting | Pancreatic Cancer | Drug: TH-302, Drug: Nab-paclitaxel, Drug: Gemcitabine | Phase 1 | 48 | EMD Serono, Threshold Pharmaceuticals |
| NCT02093962 Study of TH-302 or Placebo in Combination With Pemetrexed in Patients With Non-squamous Non-small Cell Lung Cancer | TH-CR-415 | March 2014 | July 2016 | Recruiting | Non-small Cell Lung Cancer | Drug: TH-302 combination with pemetrexed, Drug: Matched placebo in combination with pemetrexed | Phase 2 | 440 | Threshold Pharmaceuticals, EMD Serono |
| Results:
18 November 2014[34]
| |||||||||
| NCT02255110 A Japanese Trial of TH-302 in Subjects With Locally Advanced Unresectable or Metastatic Soft Tissue Sarcoma | EMR200592-008 | October 2014 | October 2016 | Recruiting | Soft Tissue Sarcoma | Drug: TH-302, Drug: Doxorubicin | Phase 2 | 46 | Merck KGaA, Threshold Pharmaceuticals |
| NCT01485042 Dose Escalation Study of Pazopanib Plus TH-302 | PATH, TH-IST-4001, Pro00031123 | December 2011 | December 2016 | Active, not recruiting | Solid Tumors | Drug: Pazopanib and TH-302 | Phase 1 | 50 | Herbert Hurwitz, MD, National Comprehensive Cancer Network, GlaxoSmithKline, Threshold Pharmaceuticals, Duke University |
| EudraCT: 2014-004072-30 A phase II trial to assess the activity and safety of TH-302 in combination with sunitinib in patients with well- and moderately-differentiated metastatic pancreatic neuroendocrine tumors (pNET) previously untreated |
GETNE-1408, EMR 200592-012 | January 2015 | Ongoing | Metastatic pancreatic neuroendocrine tumors (pNET) | Drug: TH-302 in combination with sunitinib | Phase 2 | 43 | Grupo Español de Tumores Neuroendocrinos, MFAR, S.L., Spain | |
| NCT02342379 TH-302 in Combination With Bevacizumab for Glioblastoma | TH-IST-4008, CTRC 12-0105 | February 2015 | February 2017 | Not yet recruiting | Glioblastoma | Drug: Bevacizumab and TH-302 | Phase 2 | 33 | The University of Texas Health Science Center at San Antonio |
| Note: Results based on Response Evaluation Criteria in Solid Tumors (RECIST) assessments are not shown. | |||||||||
Soft tissue sarcoma
Both, evofosfamide and ifosfamide have been investigated in combination with doxorubicin in patients with advanced soft tissue sarcoma. The study TH-CR-403 is a single arm trial investigating evofosfamide in combination with doxorubicin.[35] The study EORTC 62012 compares doxorubicin with doxorubicin plus ifosfamide.[36] Doxorubicin and ifosfamide are generic products sold by many manufacturers.
The indirect comparison of both studies shows comparable hematologic toxicity and efficacy profiles of evofosfamide and ifosfamide in combination with doxorubicin. However, a longer overall survival of patients treated with evofosfamide/doxorubicin (TH-CR-403) trial was observed. The reason for this increase is probably the increased number of patients with certain sarcoma subtypes in the evofosfamide/doxorubicin TH-CR-403 trial, see table below.
However, in the Phase 3 TH-CR-406/SARC021 study (conducted in collaboration with the Sarcoma Alliance for Research through Collaboration (SARC)), patients with locally advanced unresectable or metastatic soft tissue sarcoma treated with evofosfamide in combination with doxorubicin did not demonstrate a statistically significant improvement in OS compared with doxorubicin alone (HR: 1.06; 95% CI: 0.88 - 1.29).
| Comparison of ifosfamide/doxorubicin with evofosfamide/doxorubicin in soft tissue sarcoma | ||||
|---|---|---|---|---|
| Baseline Characteristics | Study ID | NCT00061984 Phase 3 trial EORTC 62012[37] | NCT01440088 Phase 2 trial TH-CR-403[38] | |
| Drug 1 | Doxorubicin 75 mg/m2 Day 1 or as a 72-hour continuous i.v. infusion | Doxorubicin 25 mg/m2 Days 1-3 | Doxorubicin 75 mg/m2 Day 1 | |
| Drug 2 | Ifosfamide 2.5 g/m2 Days 1-4 | Evofosfamide 300 mg/m2 Days 1, 8 | ||
| Drug 3 | Pegfilgrastim 6 mg s.c. Day 5 | Growth Factor Day 8 or Day 9 | ||
| Patients | N=228 | N=227 | N=91 | |
| Median number of cycles | 5 cycles | 6 cycles | 7 cycles (N = 91) + 10 maintenance cycles (n = 48) | |
| Number of patients with sarcoma subtype Leiomyosarcoma or Liposarcoma | 35% | 40% | 52% | |
| Efficacy | Median Progression-free survival | 4.6 months | 7.4 months | 6.5 months |
| Median Overall survival (OS) | 12.8 months | 14.3 months | 21.5 months | |
| Median OS, Leiomyosarcoma patients | 31.9 months | |||
| Median OS, Liposarcoma patients | 32.9 months | |||
| Hematologic Toxicity | Grade 3/4 Neutropenia | 37% | 42% | 31% |
| Grade 3/4 Febrile Neutropenia | 13% | 46% | 8% | |
| Grade 3/4 Thrombocytopenia | < 1% | 34% | 32% | |
| Grade 3/4 Anemia | 4% | 35% | 36% | |
| Grade 3/4 Leucopenia | 18% | 43% | N/A | |
Metastatic pancreatic cancer
Both, evofosfamide and protein-bound paclitaxel (nab-paclitaxel) have been investigated in combination with gemcitabine in patients with metastatic pancreatic cancer. The study TH-CR-404 compares gemcitabine with gemcitabine plus evofosfamide.[39] The study CA046 compares gemcitabine with gemcitabine plus nab-paclitaxel.[40] Gemcitabine is a generic product sold by many manufacturers.
The indirect comparison of both studies shows comparable efficacy profiles of evofosfamide and nab-paclitaxel in combination with gemcitabine. However, the hematologic toxicity is increased in patients treated with evofosfamide/gemcitabine (TH-CR-404 trial), see table below.
In the Phase 3 MAESTRO study, patients with previously untreated, locally advanced unresectable or metastatic pancreatic adenocarcinoma treated with evofosfamide in combination with gemcitabine did not demonstrate a statistically significant improvement in overall survival (OS) compared with gemcitabine plus placebo (hazard ratio [HR]: 0.84; 95% confidence interval [CI]: 0.71 - 1.01; p=0.0589).
| Comparison of gemcitabine/nab-paclitaxel with gemcitabine/evofosfamide in metastatic pancreatic adenocarcinoma | ||||||
|---|---|---|---|---|---|---|
| Baseline Characteristics | Study ID | NCT00844649 Phase 3 trial CA046[41] | NCT01144455 Phase 2 trial TH-CR-404[42] | |||
| Drug 1 | Gemcitabine 1 g/m2 Schedule A | Gemcitabine 1 g/m2 Schedule B | Gemcitabine 1 g/m2 Schedule B | Gemcitabine 1 g/m2 Schedule B | Gemcitabine 1 g/m2 Schedule B | |
| Drug 2 | --- | nab-Paclitaxel 125 mg/m2 Schedule B | --- | Evofosfamide 240 mg/m2 Schedule B | Evofosfamide 340 mg/m2 Schedule B | |
| Drug 3 | 15% of patients received growth factors | 26% of patients received growth factors | Optional: Growth factors | Optional: Growth factors | Optional: Growth factors | |
| Patients [N] | 430 | 431 | 69 | 71 | 74 | |
| Efficacy | Median Progression-free survival [months] | 3.7 | 5.5 | 3.6 | 5.6 | 6.0 |
| Median Overall survival [months] | 6.7 | 8.5 | 6.9 (1) | 8.7 (1) | 9.2 (1) | |
| Hematologic Toxicity | Grade 3/4 Neutropenia [%] | 27 | 38 | 17 | 34 | 43 |
| Grade 3/4 Febrile Neutropenia [%] | 1 | 3 | N/A | N/A | N/A | |
| Grade 3/4 Thrombocytopenia [%] | 9 | 13 | 12 | 30 | 55 | |
| Grade 3/4 Anemia [%] | 12 | 13 | 29 | 34 | 43 | |
| Grade 3/4 Leucopenia [%] | 16 | 31 | N/A | N/A | N/A | |
| Schedule A: Weekly for 7 of 8 weeks (cycle 1) and then on days 1, 8, and 15 every 4 weeks (cycle 2 and subsequent cycles) Schedule B: On days 1, 8 and 15 every 4 weeks | ||||||
Drug development risks
Risks published in the quarterly/annual reports of Threshold and Merck KGaA that could affect the further development of evofosfamide (TH-302):
Risks related to the formulation
The evofosfamide formulation that Threshold and Merck KGaA are using in the clinical trials was changed in 2011[43] to address issues with storage and handling requirements that were not suitable for a commercial product. Additional testing is ongoing to verify if the new formulation is suitable for a commercial product. If this new formulation is also not suitable for a commercial product another formulation has to be developed and some or all respective clinical phase 3 trials may be required to be repeated which could delay the regulatory approvals.[44]
Risks related to reimbursement
Even if Threshold/Merck KGaA succeed in obtaining regulatory approvals and bringing evofosfamide to the market, the amount reimbursed for evofosfamide may be insufficient and could adversely affect the profitability of both companies. Obtaining reimbursement for evofosfamide from third-party and governmental payors depend upon a number of factors, e.g. effectiveness of the drug, suitable storage and handling requirements of the drug and advantages over alternative treatments.
There could be the case that the data generated in the clinical trials are sufficient to obtain regulatory approvals for evofosfamide but the use of evofosfamide has a limited benefit for the third-party and governmental payors. In this case Threshold/Merck KGaA could be forced to provide supporting scientific, clinical and cost effectiveness data for the use of evofosfamide to each payor. Threshold/Merck KGaA may not be able to provide data sufficient to obtain reimbursement.[45]
Risks related to competition
Each cancer indication has a number of established medical therapies with which evofosfamide will compete, for example:
- If approved for commercial sale for pancreatic cancer, evofosfamide would compete with gemcitabine (Gemzar), marketed by Eli Lilly and Company; erlotinib (Tarceva), marketed by Genentech and Astellas Oncology; protein-bound paclitaxel (Abraxane), marketed by Celgene; and FOLFIRINOX, which is a combination of generic products that are sold individually by many manufacturers.
- If approved for commercial sale for soft tissue sarcoma, evofosfamide could potentially compete with doxorubicin or the combination of doxorubicin and ifosfamide, generic products sold by many manufacturers.[46]
Risks related to manufacture and supply
Threshold relies on third-party contract manufacturers for the manufacture of evofosfamide to meet its and Merck KGaA’s clinical supply needs. Any inability of the third-party contract manufacturers to produce adequate quantities could adversely affect the clinical development and commercialization of evofosfamide. Furthermore, Threshold has no long-term supply agreements with any of these contract manufacturers and additional agreements for more supplies of evofosfamide will be needed to complete the clinical development and/or commercialize it. In this regard, Merck KGaA has to enter into agreements for additional supplies or develop such capability itself. The clinical programs and the potential commercialization of evofosfamide could be delayed if Merck KGaA is unable to secure the supply.[47]
History
| Date | Event |
|---|---|
| Jun 2005 | Threshold files evofosfamide (TH-302) patent applications in the U.S.[48] |
| Jun 2006 | Threshold files an evofosfamide (TH-302) patent application in the EU and in Japan[49] |
| Sep 2011 | Threshold starts a Phase 3 trial (TH-CR-406) of evofosfamide in combination with doxorubicin in patients with soft tissue sarcoma |
| Feb 2012 | Threshold signs an agreement with Merck KGaA to co-develop evofosfamide |
| Apr 2012 | A Phase 2b trial (TH-CR-404) of evofosfamide in combination with gemcitabine in patients with pancreatic cancer meets primary endpoint |
| Jan 2013 | Merck KGaA starts a global Phase 3 trial (MAESTRO) of evofosfamide in combination with gemcitabine in patients with pancreatic cancer |
| Dec 2015 | two Phase 3 trials fail, Merck will not apply for a license |
References
- ↑ WHO Drug Information; Recommended INN: List 73
- ↑ Adopted Names of the United States Adopted Names Council
- ↑ Duan J; Jiao, H; Kaizerman, J; Stanton, T; Evans, JW; Lan, L; Lorente, G; Banica, M; et al. (2008). "Potent and Highly Selective Hypoxia-Activated Achiral Phosphoramidate Mustards as Anticancer Drugs". J. Med. Chem. 51 (8): 2412–20. doi:10.1021/jm701028q. PMID 18257544.
- ↑ Threshold Pharmaceuticals and Merck KGaA Announce Global Agreement to Co-Develop and Commercialize Phase 3 Hypoxia-Targeted Drug TH-302 - Press release from 3 February 2012
- ↑ Threshold Pharmaceuticals Form 8-K from 3 Nov 2014
- ↑ Weiss, G.J., Infante, J.R., Chiorean, E.G., Borad, M.J., Bendell, J.C., Molina, J.R., Tibes, R., Ramanathan, R.K., Lewandowski, K., Jones, S.F., Lacouture, M.E., Langmuir, V.K., Lee, H., Kroll, S., Burris, H.A. (2011) Phase 1 Study of the Safety, Tolerability, and Pharmacokinetics of TH-302, a Hypoxia-Activated Prodrug, in Patients with Advanced Solid Malignancies. Clinical Cancer Research 17, 2997–3004. doi:10.1158/1078-0432.CCR-10-3425
- ↑ J. Thomas Pento (2011). "TH-302". Drugs of the Future. 36 (9): 663–667. doi:10.1358/dof.2011.036.09.1678337.
- ↑ Duan J; Jiao, H; Kaizerman, J; Stanton, T; Evans, JW; Lan, L; Lorente, G; Banica, M; et al. (2008). "Potent and Highly Selective Hypoxia-Activated Achiral Phosphoramidate Mustards as Anticancer Drugs". J. Med. Chem. 51 (8): 2412–20. doi:10.1021/jm701028q. PMID 18257544.
- ↑ CPhI.cn: Synthetic routes to explore anti-pancreatic cancer drug Evofosfamide, 22 Jan 2015
- ↑ Synthetic route Reference: International patent application WO2007002931A2
- ↑ FDA Advisory Committee Briefing Materials Available for Public Release, TH-302: Pediatric oncology subcommittee of the oncologic drugs advisory committee (ODAC) meeting, December 4, 2012
- ↑ AAPS 2014 – Measurement of Diethylhexyl Phthalate (DEHP) Leached from Polyvinyl Chloride (PVC) Containing Plastics by Infusion Solutions Containing an Organic Parenteral Formulation – Poster W4210, Nov 5, 2014
- ↑ ClinicalTrials.gov
- ↑ The Primary Completion Date is defined as the date when the final subject was examined or received an intervention for the purposes of final collection of data for the primary outcome.
- ↑ Detailed Results From Positive Phase 2b Trial of TH-302 in Pancreatic Cancer at AACR Annual Meeting - Press release from 30 March 2012
- ↑ TH-302 Plus Gemcitabine vs. Gemcitabine in Patients with Untreated Advanced Pancreatic Adenocarcinoma. Borad et al. Presentation at the European Society for Medical Oncology (ESMO) 2012 Congress, September 2012. (Abstract 6660)
- ↑ Stifel 2014 Healthcare Conference; Speaker: Harold Selick - 18 November 2014
- ↑ Updated Phase 2 Results Including Analyses of Maintenance Therapy With TH-302 Following Induction Therapy With TH-302 Plus Doxorubicin in Soft Tissue Sarcoma - Press release from 15 November 2012
- ↑ TH-302 Maintenance Following TH-302 Plus Doxorubicin Induction: The Results pf a Phase 2 Study of TH-302 in Combination with Doxorubicin in Soft Tissue Sarcoma. Ganjoo et al. Connective Tissue Oncology Society (CTOS) 2012 Meeting, November 2012
- ↑ Chawla, S.P., Cranmer, L.D., Van Tine, B.A., Reed, D.R., Okuno, S.H., Butrynski, J.E., Adkins, D.R., Hendifar, A.E., Kroll, S., Ganjoo, K.N., 2014. Phase II Study of the Safety and Antitumor Activity of the Hypoxia-Activated Prodrug TH-302 in Combination With Doxorubicin in Patients With Advanced Soft Tissue Sarcoma. Journal of Clinical Oncology 32, 3299–3306. doi:10.1200/JCO.2013.54.3660
- ↑ Follow-Up Data From a Phase 1/2 Clinical Trial of TH-302 in Solid Tumors - Press release from 12 October 2010
- ↑ TH-302 Continues to Demonstrate Promising Activity in Pancreatic Cancer Phase 1/2 Clinical Trial - Press release from 24 January 2011
- ↑ TH-302, a tumor selective hypoxia-activated prodrug, complements the clinical benefits of gemcitabine in first line pancreatic cancer. Borad et al. ASCO Gastrointestinal Cancers Symposium, January 2011
- ↑ Stifel 2014 Healthcare Conference; Speaker: Harold Selick - 18 November 2014
- ↑ Borad et al., ESMO Annual Meeting, October 2010
- ↑ Video interview of Stefan Oschmann, CEO Pharma at Merck - Merck Serono Investor & Analyst Day 2014 - 18 Sept 2014 - 2:46 min - Youtube
- ↑ The Phase 3 Trial of TH-302 in Patients With Advanced Soft Tissue Sarcoma Will Continue as Planned Following Protocol-Specified Interim Analysis - Press release from 22 September 2014
- ↑ Threshold Pharmaceuticals' Partner Merck KGaA, Darmstadt, Germany, Completes Target Enrollment in the TH-302 Phase 3 MAESTRO Study in Patients With Locally Advanced or Metastatic Pancreatic Adenocarcinoma - Press release from 3 November 2014
- ↑ Data From Ongoing Phase 1/2 Trial of TH-302 Plus Bevacizumab (Avastin(R)) in Patients With Recurrent Glioblastoma - Press release from 30 May 2014
- ↑ Phase 1/2 Study of Investigational Hypoxia-Targeted Drug, TH-302, and Bevacizumab in Recurrent Glioblastoma Following Bevacizumab Failure. Brenner, et al. 2014 ASCO, 7 – 30 May 2014
- ↑ Phase 1/2 Interim Data Signaling Activity of TH-302 Plus Bevacizumab (Avastin(R)) in Patients With Glioblastoma - Press release from 17 November 2014
- ↑ Threshold Pharmaceuticals' Partner Merck KGaA, Darmstadt, Germany, Completes Target Enrollment in the TH-302 Phase 3 MAESTRO Study in Patients With Locally Advanced or Metastatic Pancreatic Adenocarcinoma - Press release from 3 November 2014
- ↑ Stifel 2014 Healthcare Conference; Speaker: Harold Selick - 18 November 2014
- ↑ Stifel 2014 Healthcare Conference; Speaker: Harold Selick - 18 November 2014
- ↑ Chawala SP, et al. J Clin Oncol. 2014 (54) 3660 doi: 10.1200/JCO.2013.54.3660
- ↑ Judson I, et al. Lancet Oncol. 2014 Apr;15(4):415-23 doi: 10.1016/S1470-2045(14)70063-4
- ↑ Judson I, et al. Lancet Oncol. 2014 Apr;15(4):415-23 doi: 10.1016/S1470-2045(14)70063-4
- ↑ Chawala SP, et al. J Clin Oncol. 2014 (54) 3660 doi: 10.1200/JCO.2013.54.3660
- ↑ Borad, M. J. et al. Randomized Phase II Trial of Gemcitabine Plus TH-302 Versus Gemcitabine in Patients With Advanced Pancreatic Cancer. Journal of Clinical Oncology (2014). doi: 10.1200/JCO.2014.55.7504
- ↑ Von Hoff, D. D. et al. Increased Survival in Pancreatic Cancer with nab-Paclitaxel plus Gemcitabine. New England Journal of Medicine 369, 1691–1703 (2013). doi: 10.1056/NEJMoa1304369
- ↑ Von Hoff, D. D. et al. Increased Survival in Pancreatic Cancer with nab-Paclitaxel plus Gemcitabine. New England Journal of Medicine 369, 1691–1703 (2013). doi: 10.1056/NEJMoa1304369
- ↑ Borad, M. J. et al. Randomized Phase II Trial of Gemcitabine Plus TH-302 Versus Gemcitabine in Patients With Advanced Pancreatic Cancer. Journal of Clinical Oncology (2014). doi: 10.1200/JCO.2014.55.7504
- ↑ Threshold Pharmaceuticals 10-K Annual report 2011 from 15 Mar 2012
- ↑ Threshold Pharmaceuticals 10-Q Quarterly report Q3/2014 from 3 Nov 14
- ↑ Threshold Pharmaceuticals Form 8-K from 9 Oct 14
- ↑ Threshold Pharmaceuticals Form 8-K from 9 Oct 14
- ↑ Threshold Pharmaceuticals Form 8-K from 9 Oct 14
- ↑ Phosphoramidate alkylator prodrugs US8003625B2, US8507464B2, US8664204B2
- ↑ Phosphoramidate alkylator prodrugs EP1896040B1 and JP5180824B2