Alkane stereochemistry
Alkane stereochemistry concerns the stereochemistry of alkanes. Alkane conformers are one of the subjects of alkane stereochemistry.
Conformation
Alkane conformers arise from rotation around sp3 hybridised carbon carbon sigma bonds. The smallest alkane with such a chemical bond, ethane, exists as an infinite number of conformations with respect to rotation around the C–C bond. Two of these are recognised as energy minimum (staggered conformation) and energy maximum (eclipsed conformation) forms. The existence of specific conformations is due to hindered rotation around sigma bonds, although a role for hyperconjugation is proposed by a competing theory.
The importance of energy minimum and energy maximum is seen by extension of these concepts to more complex molecules for which stable conformations may be predicted as minimum energy forms. The determination of stable conformations has also played a large role in the establishment of the concept of asymmetric induction and the ability to predict the stereochemistry of reactions controlled by steric effects.
In the example of staggered ethane in Newman projection, a hydrogen atom on one carbon atom has a 60° torsional angle or torsion angle [1] with respect to the nearest hydrogen atom on the other carbon so that steric hindrance is minimised. The staggered conformation is more stable by 12.5 kJ/mol than the eclipsed conformation, which is the energy maximum for ethane. In the eclipsed conformation the torsional angle is minimised.
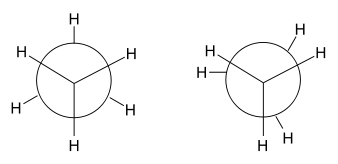
In butane, the two staggered conformations are no longer equivalent and represent two distinct conformers:the anti-conformation (left-most, below) and the gauche conformation (right-most, below).
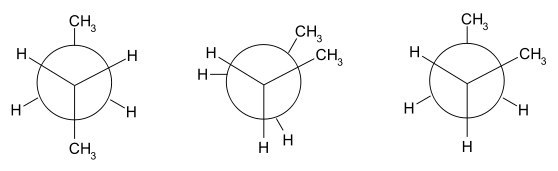
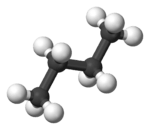
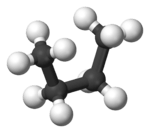
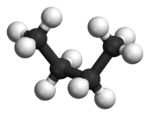
Both conformations are free of torsional strain, but, in the gauche conformation, the two methyl groups are in closer proximity than the sum of their van der Waals radii. The interaction between the two methyl groups is repulsive (van der Waals strain), and an energy barrier results.
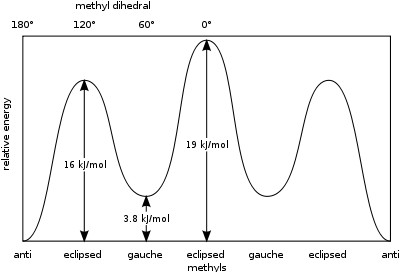
A measure of the potential energy stored in butane conformers with greater steric hindrance than the 'anti'-conformer ground state is given by these values:[2]
- Gauche, conformer – 3.8 kJ/mol
- Eclipsed H and CH3 – 16 kJ/mol
- Eclipsed CH3 and CH3 – 19 kJ/mol.
The eclipsed methyl groups exert a greater steric strain because of their greater electron density compared to lone hydrogen atoms.
The textbook explanation for the existence of the energy maximum for an eclipsed conformation in ethane is steric hindrance, but, with a C-C bond length of 154 pm and a Van der Waals radius for hydrogen of 120 pm, the hydrogen atoms in ethane are never in each other's way. The question of whether steric hindrance is responsible for the eclipsed energy maximum is a topic of debate to this day. One alternative to the steric hindrance explanation is based on hyperconjugation as analyzed within the Natural Bond Orbital framework.[3][4][5] In the staggered conformation, one C-H sigma bonding orbital donates electron density to the antibonding orbital of the other C-H bond. The energetic stabilization of this effect is maximized when the two orbitals have maximal overlap, occurring in the staggered conformation. There is no overlap in the eclipsed conformation, leading to a disfavored energy maximum. On the other hand, an analysis within quantitative molecular orbital theory shows that 2-orbital-4-electron (steric) repulsions are dominant over hyperconjugation.[6] A valence bond theory study also emphasizes the importance of steric effects.[7]
Nomenclature
Naming alkanes per standards listed in the IUPAC Gold Book is done according to the Klyne–Prelog system for specifying angles (called either torsional or dihedral angles) between substituents around a single bond:[8]
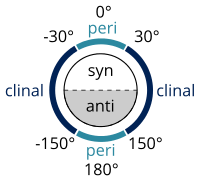
- a torsion angle between 0° and ± 90° is called syn (s)
- a torsion angle between ± 90° and 180° is called anti (a)
- a torsion angle between 30° and 150° or between –30° and –150° is called clinal
- a torsion angle between 0° and ± 30° or ± 150° and 180° is called periplanar (p)
- a torsion angle between 0° and ± 30° is called synperiplanar or syn- or cis-conformation (sp)
- a torsion angle between 30° to 90° and –30° to –90° is called synclinal or gauche or skew (sc)[9]
- a torsion angle between 90° and 150° or –90° and –150° is called anticlinal (ac)
- a torsion angle between ± 150° and 180° is called antiperiplanar or anti- or trans-conformation (ap).
Torsional strain results from resistance to twisting about a bond.
Special cases
In n-pentane, the terminal methyl groups experience additional pentane interference.
Replacing hydrogen by fluorine in polytetrafluoroethylene changes the stereochemistry from the zigzag geometry to that of a helix due to electrostatic repulsion of the fluorine atoms in the 1,3 positions. Evidence for the helix structure in the crystalline state is derived from X-ray crystallography and from NMR spectroscopy and circular dichroism in solution.[10]
See also
- More alkane conformations exist in cyclic alkanes; see cyclohexane conformations.
- More on the impact of gauche interactions; see Gauche effect.
References
- ↑ Gold Book Link
- ↑ Organic Chemistry 6e, McMurray, J.E., Brooks Cole (2003)
- ↑ Hyperconjugation not steric repulsion leads to the staggered structure of ethane. Pophristic, V. & Goodman, L. Nature 411, 565–568 (2001)Abstract
- ↑ Chemistry: A new twist on molecular shape Frank Weinhold Nature 411, 539–541 (31 May 2001)
- ↑ Rebuttal to the Bickelhaupt–Baerends case for steric repulsion causing the staggered conformation of ethane. Weinhold, F. Angew. Chem. Int. Ed. 42, 4188–4194 (2003)
- ↑ The case for steric repulsion causing the staggered conformation of ethane. Bickelhaupt, F.M. & Baerends, E.J. Angew. Chem. Int. Ed. 42, 4183–4188 (2003)
- ↑ The magnitude of hyperconjugation in ethane: A perspective from ab initio valence bond theory. Mo, Y.R. et al. Angew. Chem. Int. Ed. 43, 1986–1990 (2004)
- ↑ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "torsion angle".
- ↑ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "gauche".
- ↑ Conformational Analysis of Chiral Helical Perfluoroalkyl Chains by VCD Kenji Monde, Nobuaki Miura, Mai Hashimoto, Tohru Taniguchi, and Tamotsu Inabe J. Am. Chem. Soc.; 2006; 128(18) pp 6000–6001; Graphical abstract