Powder mixture
A powder is an assembly of dry particles dispersed in air. If two different powders are mixed perfectly, three theoretical powder mixtures can be obtained: the random mixture, the ordered mixture or the interactive mixture.
Different powder types
A powder can be free-flowing if the particles do not stick together or cohesive if the particles cling to one another to form aggregates. The likelihood of cohesion increases with decreasing size of the powder particles; particles smaller than 100 µm are generally cohesive.[1][2]
Random mixture
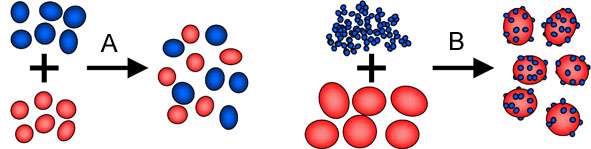
A random mixture can be obtained if two different free-flowing powders of approximately the same particle size, density and shape are mixed (see figure A).[3] Only primary particles are present in this type of mixture, i.e., the particles are not cohesive and do not cling to one another. The mixing time will determine the quality of the random mixture. However, if powders with particles of different size, density or shape are mixed the segregation can occur. Segregation will cause separation of the powders as, for example, lighter particles will be prone to travel to the top of the mixture whereas heavier particles are kept at the bottom.
Ordered mixture
The term ordered mixture was first introduced to describe a completely homogeneous mixture where the two components adhere to each other to form ordered units.[4] However, a completely homogeneous mixture is only achievable in theory and other denotations were introduced later such as adhesive mixture or interactive mixture.
Interactive mixture
If a free-flowing powder is mixed with a cohesive powder an interactive mixture can be obtained. The cohesive particles adhere to the free-flowing particles (now called carrier particles) to form interactive units as shown in figure B.[3] An interactive mixture may not contain free aggregates of the cohesive powder, which means that all small particles must be adhered to the larger ones. The difference from an ordered mixture is instead that all carrier particles do not need to be of the same size and a different number of small particles attached to each one. A narrow size range of the carrier particles is preferred to avoid segregation of the interactive units.[5] In practice a combination of a random mixture and an interactive mixture may be obtained which consists of carrier particles, aggregates of the small particles and interactive units.[6]
Formation
The formation of interactive mixtures cannot automatically be assumed, especially if smaller carrier particles [7] or a greater proportion of fine particles [8][9] are used. If an interactive mixture is to be formed, it is necessary that enough force is exerted by the carrier particles during dry mixing to break up the aggregates formed by the fine particles. Adhesion can then be achieved if the adhesive forces exceed the gravitational forces that otherwise lead to separation of the constituents.[3]
Applications
Interactive mixtures for example can be used in the manufacturing of tablets[10] enhancing the dissolution of poorly soluble drugs [11] or for nasal administration.[3] One common application is for inhalation therapy, where the concept has been used in the development of alternatives to pressurised metered dose inhalers.[12]
The schematic formation of a random mixture (A) and an interactive mixture (B) by dry mixing two powder materials.
References
- ↑ J. Bridgewater (1976). "Fundamental powder mixing mechanisms". Powder Technol. 15. 15 (2): 215–236. doi:10.1016/0032-5910(76)80051-4.
- ↑ N.A. Orr & E. Shotton (1973). "The mixing of cohesive powders". Chemical engineer. 269: 12–18.
- 1 2 3 4
- ↑ J.A. Hersey (1975). "Ordered mixing: a new concept in powder mixing practice". Powder Technol. 11: 41–44. doi:10.1016/0032-5910(75)80021-0.
- ↑ C.W. Yip & J.A. Hersey (1977). "Segregation in ordered powder mixtures". Powder Technol. 16: 149–150. doi:10.1016/0032-5910(77)85034-1.
- ↑ J.N. Staniforth (1981). "Total mixing". Int J Pharm Tech & Prod Mfr. 2: 7–12.
- ↑ C.C. Yeung & J.A. Hersey (1979). "Ordered powder mixing of coarse and fine pariculate systems". Powder Technol. 22: 127–131. doi:10.1016/0032-5910(79)85015-9.
- ↑ P. Kulvanich & P.J. Stewart (1987). "The effect of particle size and concentration on the adhesive characteristics of a model drug-carrier interactive system". J Pharm Pharmacol. 39 (9): 673–678. doi:10.1111/j.2042-7158.1987.tb06968.x. PMID 2890731.
- ↑ A.H. de Boer; P.P. Le Brun; H.G. van der Woude; P. Hagedoorn; H.G. Heijerman & H.W. Frijlink (2002). "Dry powder inhalation of antibiotics in cystic fibrosis therapy, part 1: development of a powder formulation with colistin sulfate for a special test inhaler with an air classifier as de-agglomeration principle". Eur J Pharm Biopharm. 54 (1): 17–24. doi:10.1016/S0939-6411(02)00043-7. PMID 12084498.
- ↑ Bredenberg, Susanne New Concepts in Administration of Drugs in Tablet Form : Formulation and Evaluation of a Sublingual Tablet for Rapid Absorption, and Presentation of an Individualised Dose Administration System. Doctoral thesis (2003). Uppsala University, Medicinska vetenskapsområdet, Faculty of Pharmacy, Department of Pharmacy
- ↑ C. Nystrom & M. Westerberg (1986). "The use of ordered mixtures for improving the dissolution rate of low solubility compounds". J Pharm Pharmacol. 38 (3): 161–165. doi:10.1111/j.2042-7158.1986.tb04537.x. PMID 2871148.
- ↑ H. Steckel and B. Müller. In vitro evaluation of dry powder inhalers I: drug deposition of commonly used devices (1997). "In vitro evaluation of dry powder inhalers I: Drug deposition of commonly used devices". Int J Pharm. 154: 19–29. doi:10.1016/S0378-5173(97)00113-0.