Osmium dioxide
 | |
| Names | |
|---|---|
| IUPAC name
Osmium dioxide | |
| Other names
Osmium(IV) oxide | |
| Identifiers | |
| 12036-02-1 | |
| PubChem | 187574 |
| Properties | |
| OsO2 | |
| Molar mass | 222.229 g/mol |
| Appearance | black or yellow brown |
| Density | 11.4 g/cm3 |
| Melting point | 500 °C (932 °F; 773 K) (decomposes) |
| Related compounds[1] | |
| Osmium tetroxide | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
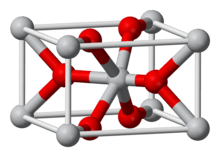
Osmium dioxide is an inorganic compound with the formula OsO2. It exists as brown to black crystalline powder, but single crystals are golden and exhibit metallic conductivity. The compound crystallizes in the rutile structural motif, i.e. the connectivity is very similar to that in the mineral rutile.
Preparation
OsO2 can be obtained by the reaction of osmium with a variety of oxidizing agents, including, sodium chlorate, osmium tetroxide, and nitric oxide at about 600 °C.[2][3] Using chemical transport, one can obtain large crystals of OsO2, sized up to 7x5x3 mm3. Single crystals show metallic resistivity of ~15 μΩ cm. A typical transport agent is O
2 via the reversible formation of volatile OsO4:[4]
- OsO2 + O2 ⇌ OsO4
Reactions
OsO2 does not dissolve in water but is attacked by dilute hydrochloric acid.[5][6] The crystals have rutile structure.[7] Unlike osmium tetroxide, OsO2 is not toxic.[8]
References
- ↑ OsO2 at webelements
- ↑ A. F. Holleman & E. Wiberg (2001). Inorganic chemistry. Academic Press. p. 1465. ISBN 0-12-352651-5.
- ↑ Thiele G.; Woditsch P. (1969). "Neutronenbeugungsuntersuchungen am Osmium(IV)-oxid". Journal of the Less Common Metals. 17 (4): 459. doi:10.1016/0022-5088(69)90074-5.
- ↑ Rogers, D. B.; Butler, S. R.; Shannon, R. D. (1972). "Single Crystals of Transition-Metal Dioxides". Inorganic Syntheses. XIV: 135–145. doi:10.1002/9780470132449.ch27.
- ↑ J. E. Greedan; D. B. Willson; T. E. Haas (1968). "Metallic nature of osmium dioxide". Inorg. Chem. 7 (11): 2461–2463. doi:10.1021/ic50069a059.
- ↑ Yen, P (2004). "Growth and characterization of OsO
2 single crystals". Journal of Crystal Growth. 262 (1-4): 271. doi:10.1016/j.jcrysgro.2003.10.021. - ↑ Boman C.E.; Danielsen, Jacob; Haaland, Arne; Jerslev, Bodil; Schäffer, Claus Erik; Sunde, Erling; Sørensen, Nils Andreas (1970). "Precision Determination of the Crystal Structure of Osmium Dioxide". Acta Chemica Scandinavica. 24: 123–128. doi:10.3891/acta.chem.scand.24-0123.
- ↑ Smith, I.C., B.L. Carson, and T.L. Ferguson (1974). "Osmium: An appraisal of environmental exposure.". Env Health Perspect. Brogan &. 8: 201–213. doi:10.2307/3428200. JSTOR 3428200. PMC 1474945
 . PMID 4470919.
. PMID 4470919.