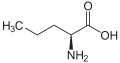
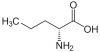Norvaline
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
2-Aminopentanoic acid | |||
| Other names
2-Aminopentyric acid; α-Aminopentanoic acid; Propylglycine | |||
| Identifiers | |||
| 6600-40-4 | |||
| 3D model (Jmol) | (L): Interactive image (DL): Interactive image (D): Interactive image | ||
| ChEMBL | ChEMBL55612 | ||
| ChemSpider | 58608 (L) 801 (DL) 388660 (D) | ||
| ECHA InfoCard | 100.026.858 | ||
| PubChem | 65098 | ||
| |||
| |||
| Properties | |||
| C5H11NO2 | |||
| Molar mass | 117.15 g·mol−1 | ||
| Acidity (pKa) | 2.36 (carboxyl), 9.76 (amino)[1] | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Norvaline (abbreviated as Nva) is an amino acid with the formula CH3(CH2)2CH(NH2)CO2H. The compound is an isomer of the more common amino acid valine.[2] Like most other α-amino acids, norvaline is chiral. It is a white, water-soluble solid.
Occurrence
Norvaline is a non-proteinogenic unbranched-chain amino acid. It has previously been reported to be a natural component of an antifungal peptide of Bacillus subtilis. Norvaline and other modified unbranched chain amino acids have received attention because they appear to be incorporated in some recombinant proteins found in E. coli.[3] Its biosynthesis has been examined. The incorporation of Nva into peptides reflects the imperfect selectivity of the associated aminoacyl-tRNA synthetase. In Miller–Urey experiments probing prebiotic synthesis of amino acids, norvaline, but also norleucine, are produced.[4]
Norvaline is known to promote tissue regeneration and muscle growth,[5] and to become a precursor in the penicillin biosynthetic pathway.[6][7]
Nomenclature
Norvaline and norleucine (one hydrocarbon group longer) both possess the nor- prefix for historical reason, despite current conventional usage of the prefix to denote a missing hydrocarbon group (under which they would theoretically be called "dihomoalanine" and "trihomoalanine"). The name is not systematic.[8]
References
- ↑ Dawson, R.M.C., et al., Data for Biochemical Research, Oxford, Clarendon Press, 1959.
- ↑ Merriam-Webster Retrieved 4 September 2010
- ↑ Soini J, Falschlehner C, Liedert C, Bernhardt J, Vuoristo J, Neubauer P (2008). "Norvaline is accumulated after a down-shift of oxygen in Escherichia coli W3110". Microbial Cell Factories. 7: 30. doi:10.1186/1475-2859-7-30. PMC 2579280
 . PMID 18940002.
. PMID 18940002. - ↑ Alvarez-Carreño C, Becerra A, Lazcano A (October 2013). "Norvaline and norleucine may have been more abundant protein components during early stages of cell evolution". Origins of Life and Evolution of the Biosphere. 43 (4-5): 363–75. doi:10.1007/s11084-013-9344-3. PMID 24013929.
- ↑ Ming XF, Rajapakse AG, Carvas JM, Ruffieux J, Yang Z (2009). "Inhibition of S6K1 accounts partially for the anti-inflammatory effects of the arginase inhibitor L-norvaline". BMC Cardiovascular Disorders. 9: 12. doi:10.1186/1471-2261-9-12. PMC 2664787
 . PMID 19284655.
. PMID 19284655. - ↑ reference.md Retrieved 4 September 2010
- ↑ Kisumi M, Sugiura M, Chibata I (August 1976). "Biosynthesis of norvaline, norleucine, and homoisoleucine in Serratia marcescens". Journal of Biochemistry. 80 (2): 333–9. PMID 794063.
- ↑ "Nomenclature and Symbolism For Amino Acids and Peptides". Pure and Applied Chemistry. 56 (5): 595–624. 1984. doi:10.1351/pac198456050595.