Lucifer yellow
 | |
| Names | |
|---|---|
| IUPAC name
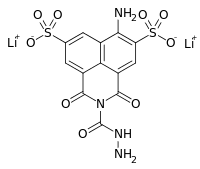
dilithium 6-Amino-2-(hydrazinecarbonyl)-1,3-dioxobenzo[de]isoquinoline-5,8-disulfonate | |
| Identifiers | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:52104 |
| ChemSpider | 20137740 |
| PubChem | 20835957 |
| |
| |
| Properties | |
| C13H10Li2N4O9S2 | |
| Molar mass | 444.24 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Lucifer yellow is a fluorescent dye used in cell biology.[1] The key property of Lucifer yellow is that it can readily visualized in both living and fixed cells using a fluorescence microscope. Lucifer yellow was invented by Walter W. Stewart at the National Institutes of Health and patented in 1978.[2]
Preparations
For common usage it is compounded with carbohydrazide (CH) and prepared as a lithium salt. The CH group allows it to be covalently linked to surrounding biomolecules during aldehyde fixation.[3]
Other cations such as ammonium or potassium can be used when lithium is undesirable, but the resulting salts are less soluble in water.
Lucifer yellow can also be compounded as a vinyl sulfone, with ethylenediamine, or with cadaverine.
References
- ↑ Hanani, Menachem (January 2012). "Lucifer yellow – an angel rather than the devil". Journal of Cellular and Molecular Medicine. 16 (2): 22–31. doi:10.1111/j.1582-4934.2011.01378.x. PMID 21740513.
- ↑ Patent description
- ↑ "Lucifer Yellow CH, Lithium Salt". Molecular Probes. Retrieved 17 March 2014.
External links
- Invitrogen's manual for Lucifer yellow
- Molecular structure and spectra of Lucifer yellow CH (lithium salt)
This article is issued from Wikipedia - version of the 6/17/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.