Kazal domain
| Kazal-type serine protease inhibitor domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
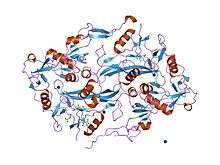 the structure of the follistatin:activin complex | |||||||||
| Identifiers | |||||||||
| Symbol | Kazal_1 | ||||||||
| Pfam | PF00050 | ||||||||
| InterPro | IPR002350 | ||||||||
| PROSITE | PDOC00254 | ||||||||
| SCOP | 3sgb | ||||||||
| SUPERFAMILY | 3sgb | ||||||||
| |||||||||
| Kazal-type serine protease inhibitor domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
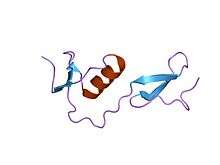 structure of fs1, the heparin-binding domain of follistatin | |||||||||
| Identifiers | |||||||||
| Symbol | Kazal_2 | ||||||||
| Pfam | PF07648 | ||||||||
| InterPro | IPR011497 | ||||||||
| PROSITE | PDOC00254 | ||||||||
| SCOP | 3sgb | ||||||||
| SUPERFAMILY | 3sgb | ||||||||
| |||||||||
The Kazal domain is an evolutionary conserved protein domain usually indicative of serine protease inhibitors. However, kazal-like domains are also seen in the extracellular part of agrins, which are not known to be protease inhibitors.
In animals, serine protease inhibitors that act via their Kazal domain are grouped under the MEROPS inhibitor family I1, clan IA.[1][2]
Kazal 1
Kazal domains often occur in tandem arrays. Small alpha+beta fold containing three disulfide bonds. Alignment also includes a single domain from transporters in the OATP/PGT family P46721.
Peptide proteinase inhibitors can be found as single domain proteins or as single or multiple domains within proteins; these are referred to as either simple or compound inhibitors, respectively. In many cases they are synthesised as part of a larger precursor protein, either as a prepropeptide or as an N-terminal domain associated with an inactive peptidase or zymogen. This domain prevents access of the substrate to the active site. Removal of the N-terminal inhibitor domain either by interaction with a second peptidase or by autocatalytic cleavage activates the zymogen. Other inhibitors interact direct with proteinases using a simple noncovalent lock and key mechanism; while yet others use a conformational change-based trapping mechanism that depends on their structural and thermodynamic properties.
This family of Kazal inhibitors, belongs to MEROPS inhibitor family I1, clan IA. They inhibit serine peptidases of the S1 family (INTERPRO).[3] The members are primarily metazoan, but includes exceptions in the alveolata (apicomplexa), stramenopiles, higher plants and bacteria.
Kazal inhibitors, which inhibit a number of serine proteases (such as trypsin and elastase), belong to family of proteins that includes pancreatic secretory trypsin inhibitor; avian ovomucoid; acrosin inhibitor; and elastase inhibitor. These proteins contain between 1 and 7 Kazal-type inhibitor repeats.[4][5]
The structure of the Kazal repeat includes a large quantity of extended chain, 2 short alpha-helices and a 3-stranded anti-parallel beta sheet.[4] The inhibitor makes 11 contacts with its enzyme substrate: unusually, 8 of these important residues are hypervariable.[5] Altering the enzyme-contact residues, and especially that of the active site bond, affects the strength of inhibition and specificity of the inhibitor for particular serine proteases.[5][6] The presence of this Pfam domain is usually indicative of serine protease inhibitors, however, Kazal-like domains are also seen in the extracellular part of agrins which are not known to be proteinase inhibitors.
Human proteins with Kazal 1 domains:
- AGRIN, CPAMD8
- FST, FSTL3, FSTL4, FSTL5
- IGFBPL1
- SMOC1, SPARC, SPARCL1, SPINK1, SPINK2, SPINK4, SPINK5, SPINK5L2, SPINK5L3, SPINK6, SPINK7, SPINK9
- TMEFF1, TMEFF2
Kazal 2
This domain is usually indicative of serine protease inhibitors that belong to Merops inhibitor families: I1, I2, I17 and I31. However, kazal-like domains are also seen in the extracellular part of agrins, which are not known to be protease inhibitors. Kazal domains often occur in tandem arrays and have a central alpha-helix, a short two-stranded antiparallel beta-sheet and several disulphide bonds.[7][8][9] The amino terminal segment of this domain binds to the active site of its target proteases, thus inhibiting their function.
Human proteins with Kazal 2 domains:
- C6, CFI
- FSTL1, FSTL3
- HTRA1, HTRA3, HTRA4
- IGFBP7, KAZALD1, LST3, RECK
- SLC21A8, SLCO1A2, SLCO1B1, SLCO1B3, SLCO1C1, SLCO2A1, SLCO3A1, SLCO4A1, SLCO4C1, SLCO5A1, SLCO6A1, SMOC2, SPINK5, SPOCK1, SPOCK2, SPOCK3
- WFIKKN1, WFIKKN2
References
- ↑ MEROPS family I1
- ↑ InterPro: IPR001239
- ↑ Rawlings ND, Tolle DP, Barrett AJ (March 2004). "Evolutionary families of peptidase inhibitors". Biochem. J. 378 (Pt 3): 705–16. doi:10.1042/BJ20031825. PMC 1224039
 . PMID 14705960.
. PMID 14705960. - 1 2 Williamson MP; Marion D; Wüthrich K (March 1984). "Secondary structure in the solution conformation of the proteinase inhibitor IIA from bull seminal plasma by nuclear magnetic resonance". J. Mol. Biol. 173 (3): 341–59. doi:10.1016/0022-2836(84)90125-6. PMID 6699915.
- 1 2 3 Laskowski M, Kato I, Ardelt W, Cook J, Denton A, Empie MW, Kohr WJ, Park SJ, Parks K, Schatzley BL (January 1987). "Ovomucoid third domains from 100 avian species: isolation, sequences, and hypervariability of enzyme-inhibitor contact residues". Biochemistry. 26 (1): 202–21. doi:10.1021/bi00375a028. PMID 3828298.
- ↑ Empie MW, Laskowski M (May 1982). "Thermodynamics and kinetics of single residue replacements in avian ovomucoid third domains: effect on inhibitor interactions with serine proteinases". Biochemistry. 21 (10): 2274–84. doi:10.1021/bi00539a002. PMID 7046785.
- ↑ Schlott B, Wöhnert J, Icke C, Hartmann M, Ramachandran R, Gührs KH, Glusa E, Flemming J, Görlach M, Grosse F, Ohlenschläger O (April 2002). "Interaction of Kazal-type inhibitor domains with serine proteinases: biochemical and structural studies". J. Mol. Biol. 318 (2): 533–46. doi:10.1016/S0022-2836(02)00014-1. PMID 12051857.
- ↑ Stubbs MT, Morenweiser R, Stürzebecher J, Bauer M, Bode W, Huber R, Piechottka GP, Matschiner G, Sommerhoff CP, Fritz H, Auerswald EA (August 1997). "The three-dimensional structure of recombinant leech-derived tryptase inhibitor in complex with trypsin. Implications for the structure of human mast cell tryptase and its inhibition". J. Biol. Chem. 272 (32): 19931–7. doi:10.1074/jbc.272.32.19931. PMID 9242660.
- ↑ van de Locht A, Lamba D, Bauer M, Huber R, Friedrich T, Kröger B, Höffken W, Bode W (November 1995). "Two heads are better than one: crystal structure of the insect derived double domain Kazal inhibitor rhodniin in complex with thrombin". EMBO J. 14 (21): 5149–57. PMC 394622
 . PMID 7489704.
. PMID 7489704.
This article incorporates text from the public domain Pfam and InterPro IPR002350 This article incorporates text from the public domain Pfam and InterPro IPR011497