Isobutane (data page)
This page provides supplementary chemical data on isobutane.
Material Safety Data Sheet
The handling of this chemical may incur notable safety precautions. It is highly recommend that you seek the Material Safety Datasheet (MSDS) for this chemical from a reliable source such as SIRI, and follow its directions.
Structure and properties
| Structure and properties | |
|---|---|
| Index of refraction, nD | ? |
| Abbe number | ? |
| Dielectric constant, εr | ? ε0 at ? °C |
| Bond strength | ? |
| Bond length | ? |
| Bond angle | ? |
| Magnetic susceptibility | ? |
| Surface tension | 10.3 dyn/cm at 20°C P' 300 kPa |
Thermodynamic properties
| Phase behavior | |
|---|---|
| Triple point | 113.55K (–159.6 °C), 0.019481 Pa |
| Critical point | 407.7 K (134.6 °C), 3650 kPa |
| Std enthalpy change of fusion, ΔfusH |
4.54 kJ/mol |
| Std entropy change of fusion, ΔfusS |
39.92 J/(mol·K) |
| Std enthalpy change of vaporization, ΔvapH |
21.3 kJ/mol |
| Std entropy change of vaporization, ΔvapS |
81.46 J/(mol·K) |
| Solid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Liquid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
200.79 J/(mol K) |
| Heat capacity, cp | 129.70 J/(mol K) –253°C to –13°C |
| Gas properties | |
| Std enthalpy change of formation, ΔfH |
–134.2 kJ/mol |
| Standard molar entropy, S |
249.7 J/(mol K) |
| Enthalpy of combustion, ΔcH |
–2869 kJ/mol |
| Heat capacity, cp | 95.21 J/(mol K) at 20°C |
| van der Waals' constants[1] | a = 1304.1 L2 kPa/mol2 b = 0.1142 liter per mole |
Vapor pressure of liquid
| P in mm Hg | 1 | 10 | 40 | 100 | 400 | 760 | 1520 | 3800 | 7600 | 15200 | 30400 | 45600 | |
| T in °C | –109.2 | –86.4 | –68.4 | –54.1 | –27.1 | –11.7 | 7.5 | 39.0 | 68.8 | 99.5 | — | — | |
Table data obtained from CRC Handbook of Chemistry and Physics 44th ed.
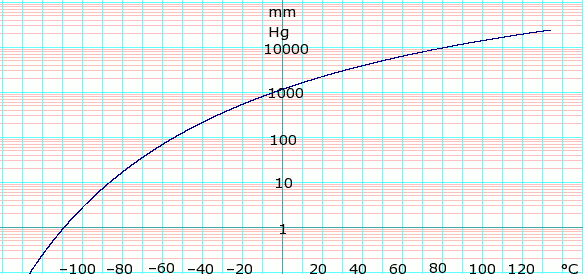
Vapor pressure of iso-butane. From formula: 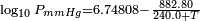 obtained from Lange's Handbook of Chemistry, 10th ed.
obtained from Lange's Handbook of Chemistry, 10th ed.
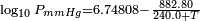 obtained from Lange's Handbook of Chemistry, 10th ed.
obtained from Lange's Handbook of Chemistry, 10th ed.Spectral data
| UV-Vis | |
|---|---|
| λmax | ? nm |
| Extinction coefficient, ε | ? |
| IR | |
| Major absorption bands | ? cm−1 |
| NMR | |
| Proton NMR | |
| Carbon-13 NMR | |
| Other NMR data | |
| MS | |
| Masses of main fragments |
|
References
- ↑ Lange's Handbook of Chemistry 10th ed, pp 1522-1524
Except where noted otherwise, data relate to standard ambient temperature and pressure.
Disclaimer applies.
This article is issued from Wikipedia - version of the 3/30/2013. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.