Interference (genetic)
Crossover interference is the term used to refer to the non-random placement of crossovers with respect to each other during meiosis. The term is attributed to Muller, who observed that one crossover "interferes with the coincident occurrence of another crossing over in the same pair of chromosomes, and I have accordingly termed this phenomenon ‘interference’."[1]
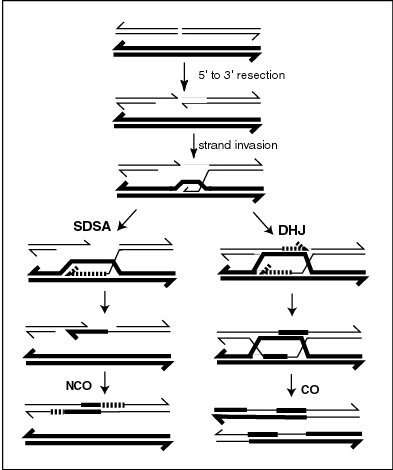
Meiotic crossovers (COs) appear to be regulated to ensure that COs on the same chromosome are distributed far apart (crossover interference). In the nematode worm Caenorhabditis elegans, meiotic double-strand breaks (DSBs) outnumber COs. Thus not all DSBs are repaired by a recombination process(es) leading to COs. The RTEL-1 protein is required to prevent excess meiotic COs. In rtel-1 mutants meiotic CO recombination is significantly increased and crossover interference appears to be absent.[2] RTEL1 likely acts by promoting synthesis-dependent strand annealing which results in non-crossover (NCO) recombinants instead of COs (see diagram).[2] Normally, about half of all DSBs are converted into NCOs. RTEL-1 appears to enforce meiotic crossover interference by directing the repair of some DSBs towards NCOs rather than COs.[2]
In humans, recombination rate increases with maternal age.[3] Furthermore, placement of female recombination events appears to become increasingly deregulated with maternal age, with a larger fraction of events occurring within closer proximity to each other than would be expected under simple models of crossover interference.[4]
References
- ↑ Muller, H.J. (1916). "The mechanism of crossing over". Am. Nat. 50.
- 1 2 3 Youds JL, Mets DG, McIlwraith MJ, Martin JS, Ward JD, ONeil NJ, Rose AM, West SC, Meyer BJ, Boulton SJ (2010). "RTEL-1 enforces meiotic crossover interference and homeostasis". Science. 327 (5970): 1254–8. doi:10.1126/science.1183112. PMID 20203049.
- ↑ Kong A, Barnard J, Gudbjartsson DF, Thorleifsson G, Jonsdottir G, Sigurdardottir S, Richardsson B, Jonsdottir J, Thorgeirsson T, Frigge ML, Lamb NE, Sherman S, Gulcher JR, Stefansson K (2004). "Recombination rate and reproductive success in humans". Nat. Genet. 36 (11): 1203–6. doi:10.1038/ng1445. PMID 15467721.
- ↑ Campbell CL, Furlotte NA, Eriksson N, Hinds D, Auton A (2015). "Escape from crossover interference increases with maternal age". Nat Commun. 6: 6260. doi:10.1038/ncomms7260. PMC 4335350
 . PMID 25695863.
. PMID 25695863.
External links
 Media related to Genetic interference at Wikimedia Commons
Media related to Genetic interference at Wikimedia Commons