Imidazolidinyl urea
 | |
 | |
| Names | |
|---|---|
| IUPAC names
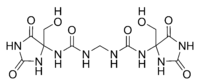
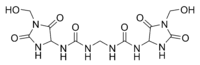
Correct new structure (upper pic.): 1,1'-methylenebis{3-[4-(hydroxymethyl)-2,5-dioxoimidazolidin-4-yl]urea} Erroneous old structure (lower pic.): 1,1'-methylenebis{3-[1-(hydroxymethyl)-2,5-dioxoimidazolidin-4-yl]urea} | |
| Other names
Imidurea, Germall 115;
N',N''-methylenebis[3-[1-(hydroxymethyl)- 2,5-dioxoimidazolidin-4-yl]urea]; 1-[1-(hydroxymethyl)-2,5-dioxoimidazolidin-4-yl]- 3-[[[1-(hydroxymethyl)-2,5-dioxoimidazolidin-4-yl] carbamoylamino]methyl]urea | |
| Identifiers | |
| 39236-46-9 | |
| 3D model (Jmol) | Interactive image Interactive image |
| ChEBI | CHEBI:51805 |
| ChEMBL | ChEMBL65433 |
| ChemSpider | 35067 |
| ECHA InfoCard | 100.049.411 |
| EC Number | 254-372-6 |
| PubChem | 38258 |
| |
| |
| Properties | |
| C11H16N8O8 | |
| Molar mass | 388.29 g/mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Imidazolidinyl urea is an antimicrobial preservative used in cosmetics . It is chemically related to diazolidinyl urea which is used in the same way. Imidazolidinyl urea acts as a formaldehyde releaser.
Safety
Some people have a contact allergy to imidazolidinyl urea causing dermatitis.[1] Such people are often also allergic to diazolidinyl urea.
Chemistry
Imidazolidinyl urea was poorly characterized until recently and the single Chemical Abstracts Service structure assigned to it is probably not the major one in the commercial material. Instead, new data indicate that the hydroxymethyl functional group of each imidazolidine ring is attached to the carbon, rather than on the nitrogen atom:[2]
 |
 |
| Originally reported structure | Hoeck's revised structure |
Synthesis
Imidazolidinyl urea is produced by the chemical reaction of allantoin and formaldehyde in the presence of sodium hydroxide solution and heat. The reaction mixture is then neutralized with hydrochloric acid and evaporated:
2  + 3 H2C=O →
+ 3 H2C=O → 
Commercial imidazolidinyl urea is a mixture of different formaldehyde addition products including polymers.[2]
References
- ↑ Review of toxicological data (NTP NIEHS)
- 1 2 Lehmann SV; Hoeck U; Breinholdt J; Olsen CE; Kreilgaard B. (2006). "Characterization and chemistry of imidazolidinyl urea and diazolidinyl urea". Contact Dermatitis. 54 (1): 50–58. doi:10.1111/j.0105-1873.2006.00735.x. PMID 16426294.