HIST1H2BM
Histone H2B type 1-M is a protein that in humans is encoded by the HIST1H2BM gene.[3][4][5]
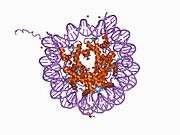
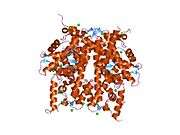
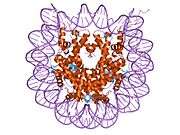
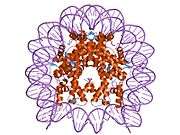
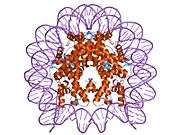
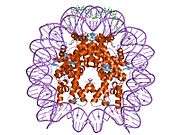
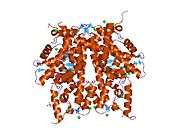
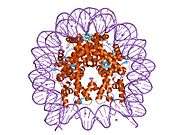
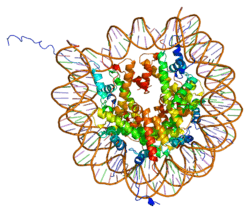
Histones are basic nuclear proteins that are responsible for the nucleosome structure of the chromosomal fiber in eukaryotes. Two molecules of each of the four core histones (H2A, H2B, H3, and H4) form an octamer, around which approximately 146 bp of DNA is wrapped in repeating units, called nucleosomes. The linker histone, H1, interacts with linker DNA between nucleosomes and functions in the compaction of chromatin into higher order structures. This gene is intronless and encodes a member of the histone H2B family. Transcripts from this gene lack polyA tails but instead contain a palindromic termination element. This gene is found in the small histone gene cluster on chromosome 6p22-p21.3.[5]
References
Further reading
- El Kharroubi A, Piras G, Zensen R, Martin MA (1998). "Transcriptional activation of the integrated chromatin-associated human immunodeficiency virus type 1 promoter.". Mol. Cell. Biol. 18 (5): 2535–44. doi:10.1128/mcb.18.5.2535. PMC 110633
 . PMID 9566873.
. PMID 9566873.
- Albig W, Trappe R, Kardalinou E, et al. (1999). "The human H2A and H2B histone gene complement.". Biol. Chem. 380 (1): 7–18. doi:10.1515/BC.1999.002. PMID 10064132.
- Deng L, de la Fuente C, Fu P, et al. (2001). "Acetylation of HIV-1 Tat by CBP/P300 increases transcription of integrated HIV-1 genome and enhances binding to core histones.". Virology. 277 (2): 278–95. doi:10.1006/viro.2000.0593. PMID 11080476.
- Deng L, Wang D, de la Fuente C, et al. (2001). "Enhancement of the p300 HAT activity by HIV-1 Tat on chromatin DNA.". Virology. 289 (2): 312–26. doi:10.1006/viro.2001.1129. PMID 11689053.
- Strausberg RL, Feingold EA, Grouse LH, et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences.". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. doi:10.1073/pnas.242603899. PMC 139241
 . PMID 12477932.
. PMID 12477932.
- Cheung WL, Ajiro K, Samejima K, et al. (2003). "Apoptotic phosphorylation of histone H2B is mediated by mammalian sterile twenty kinase.". Cell. 113 (4): 507–17. doi:10.1016/S0092-8674(03)00355-6. PMID 12757711.
- Mungall AJ, Palmer SA, Sims SK, et al. (2003). "The DNA sequence and analysis of human chromosome 6.". Nature. 425 (6960): 805–11. doi:10.1038/nature02055. PMID 14574404.
- Lusic M, Marcello A, Cereseto A, Giacca M (2004). "Regulation of HIV-1 gene expression by histone acetylation and factor recruitment at the LTR promoter.". EMBO J. 22 (24): 6550–61. doi:10.1093/emboj/cdg631. PMC 291826
 . PMID 14657027.
. PMID 14657027.
- Gerhard DS, Wagner L, Feingold EA, et al. (2004). "The status, quality, and expansion of the NIH full-length cDNA project: the Mammalian Gene Collection (MGC).". Genome Res. 14 (10B): 2121–7. doi:10.1101/gr.2596504. PMC 528928
 . PMID 15489334.
. PMID 15489334.
- Golebiowski F, Kasprzak KS (2007). "Inhibition of core histones acetylation by carcinogenic nickel(II).". Mol. Cell. Biochem. 279 (1-2): 133–9. doi:10.1007/s11010-005-8285-1. PMID 16283522.
- Zhu B, Zheng Y, Pham AD, et al. (2006). "Monoubiquitination of human histone H2B: the factors involved and their roles in HOX gene regulation.". Mol. Cell. 20 (4): 601–11. doi:10.1016/j.molcel.2005.09.025. PMID 16307923.
- Bonenfant D, Coulot M, Towbin H, et al. (2006). "Characterization of histone H2A and H2B variants and their post-translational modifications by mass spectrometry.". Mol. Cell Proteomics. 5 (3): 541–52. doi:10.1074/mcp.M500288-MCP200. PMID 16319397.
- Siuti N, Roth MJ, Mizzen CA, et al. (2006). "Gene-specific characterization of human histone H2B by electron capture dissociation.". J. Proteome Res. 5 (2): 233–9. doi:10.1021/pr050268v. PMID 16457587.
- Pavri R, Zhu B, Li G, et al. (2006). "Histone H2B monoubiquitination functions cooperatively with FACT to regulate elongation by RNA polymerase II.". Cell. 125 (4): 703–17. doi:10.1016/j.cell.2006.04.029. PMID 16713563.
- Kim SC, Sprung R, Chen Y, et al. (2006). "Substrate and functional diversity of lysine acetylation revealed by a proteomics survey.". Mol. Cell. 23 (4): 607–18. doi:10.1016/j.molcel.2006.06.026. PMID 16916647.
PDB gallery |
|---|
|
| 1aoi: COMPLEX BETWEEN NUCLEOSOME CORE PARTICLE (H3,H4,H2A,H2B) AND 146 BP LONG DNA FRAGMENT |
| 1eqz: X-RAY STRUCTURE OF THE NUCLEOSOME CORE PARTICLE AT 2.5 A RESOLUTION |
| 1hq3: CRYSTAL STRUCTURE OF THE HISTONE-CORE-OCTAMER IN KCL/PHOSPHATE |
| 1p34: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1p3a: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1p3b: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1p3f: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1p3g: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1p3i: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1p3k: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1p3l: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1p3m: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1p3o: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1p3p: Crystallographic Studies of Nucleosome Core Particles containing Histone 'Sin' Mutants |
| 1s32: Molecular Recognition of the Nucleosomal 'Supergroove' |
| 1tzy: Crystal Structure of the Core-Histone Octamer to 1.90 Angstrom Resolution |
| 1u35: Crystal structure of the nucleosome core particle containing the histone domain of macroH2A |
| 2aro: Crystal Structure Of The Native Histone Octamer To 2.1 Angstrom Resolution, Crystalised In The Presence Of S-Nitrosoglutathione |
| 2cv5: Crystal structure of human nucleosome core particle |
| 2f8n: 2.9 Angstrom X-ray structure of hybrid macroH2A nucleosomes |
| 2hio: HISTONE OCTAMER (CHICKEN), CHROMOSOMAL PROTEIN |
|
|
 . PMID 9566873.
. PMID 9566873. . PMID 12477932.
. PMID 12477932. . PMID 14657027.
. PMID 14657027. . PMID 15489334.
. PMID 15489334.