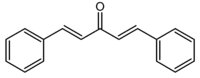
Dibenzylideneacetone
 | |
 | |
| | |
| Names | |
|---|---|
| IUPAC name
(1E, 4E)-1,5-Diphenylpenta-1,4-dien-3-one | |
| Other names
Dibenzalacetone | |
| Identifiers | |
| 538-58-9 undefined 35225-79-7 (E,E) 115587-57-0 (E,Z) 58321-78-1 (Z,Z) | |
| 3D model (Jmol) | undefined: Interactive image (E,E): Interactive image (E,Z): Interactive image (Z,Z): Interactive image |
| ChEMBL | ChEMBL17201 (E,E) |
| ChemSpider | 86113 undefined 555548 (E,E) 1266463 (E,Z) 1266462 (Z,Z) |
| ECHA InfoCard | 100.007.908 |
| PubChem | 95417 undefined 640180 (E,E) 1549622 (E,Z) 1549621 (Z,Z) |
| UNII | 9QXO7BCY9L (E,E) N4SH2VDI6Y (E,Z) 5L4OCE3E5U (Z,Z) |
| |
| |
| Properties | |
| C17H14O | |
| Molar mass | 234.29 g/mol |
| Appearance | Yellow solid |
| Melting point | *Aldrich 110-111 °C (trans, trans isomer)
|
| Boiling point | 130 °C (266 °F; 403 K) (cis, cis isomer) *Merck |
| insoluble | |
| Hazards | |
| Main hazards | Irritant |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |

Dibenzylideneacetone or dibenzalacetone, often abbreviated dba, is an organic compound with the formula C17H14O. It is a pale-yellow solid insoluble in water, but soluble in ethanol. Dibenzylideneacetone is used as a component in sunscreens and as a ligand in organometallic chemistry.
Preparation
The compound can be prepared in the laboratory through condensation of benzaldehyde and acetone with sodium hydroxide in a water/ethanol medium with the exclusive formation of the trans, trans isomer (melting point 110–111 °C).[1]
This reaction is frequently encountered in organic chemistry education as a laboratory procedure. The conversion proceeds via the intermediacy of benzylideneacetone.
Reactions and derivatives
Prolonged exposure to sunlight converts the compound in a [2+2] cycloaddition to a mixture of four cyclobutane isomers.[2]
Dibenzylideneacetone is a component of the catalyst tris(dibenzylideneacetone)dipalladium(0). It is a labile ligand that is easily displaced by triphenylphosphine, hence it serves a useful entry point into palladium(0) chemistry.
References
- ↑ Conard, C. R.; Dolliver, M. A. (1943). "Dibenzalacetone". Org. Synth.; Coll. Vol., 2, p. 167
- ↑ Rao, G. N.; Janardhana, C.; Ramanathan, V.; Rajesh, T.; Kumar, P. H. (November 2006). "Photochemical Dimerization of Dibenzylideneacetone. A Convenient Exercise in [2+2] Cycloaddition Using Chemical Ionization Mass Spectrometry". J. Chem. Educ. 83 (11): 1667. doi:10.1021/ed083p1667.
