Creatine
 | |
 | |
| Names | |
|---|---|
| Systematic IUPAC name
2-[Carbamimidoyl(methyl)amino]acetic acid | |
| Other names
N-Carbamimidoyl-N-methylglycine; Methylguanidoacetic acid | |
| Identifiers | |
| 57-00-1 | |
| 3D model (Jmol) | Interactive image Interactive image |
| 3DMet | B00084 |
| 907175 | |
| ChEBI | CHEBI:16919 |
| ChEMBL | ChEMBL283800 |
| ChemSpider | 566 |
| DrugBank | DB00148 |
| ECHA InfoCard | 100.000.278 |
| EC Number | 200-306-6 |
| 240513 | |
| KEGG | C00300 |
| MeSH | Creatine |
| PubChem | 586 |
| RTECS number | MB7706000 |
| UNII | MU72812GK0 |
| |
| |
| Properties | |
| C4H9N3O2 | |
| Molar mass | 131.14 g·mol−1 |
| Appearance | White crystals |
| Odor | Odourless |
| Melting point | 255 °C (491 °F; 528 K) |
| 13.3 g L−1 (at 18 °C) | |
| log P | −1.258 |
| Acidity (pKa) | 3.429 |
| Basicity (pKb) | 10.568 |
| Isoelectric point | 8.47 |
| Thermochemistry | |
| 171.1 J K−1 mol−1 (at 23.2 °C) | |
| Std molar entropy (S |
189.5 J K−1 mol−1 |
| Std enthalpy of formation (ΔfH |
−538.06–−536.30 kJ mol−1 |
| Std enthalpy of combustion (ΔcH |
−2.3239–−2.3223 MJ mol−1 |
| Pharmacology | |
| C01EB06 (WHO) | |
| Pharmacokinetics: | |
| 3 hours | |
| Hazards | |
| GHS pictograms |  |
| GHS signal word | WARNING |
| H315, H319, H335 | |
| P261, P305+351+338 | |
| EU classification (DSD) |
|
| R-phrases | R36/37/38 |
| S-phrases | S26, S36 |
| Related compounds | |
| Related alkanoic acids |
|
| Related compounds |
Dimethylacetamide |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Creatine (/ˈkriːətiːn/ or /ˈkriːətɪn/[1][2]) is a nitrogenous organic acid that occurs naturally in vertebrates and helps to supply energy to all cells in the body, primarily muscle. This is achieved by increasing the formation of adenosine triphosphate (ATP). Early analysis showed that human blood is approximately 1% creatine, and the highest concentrations are found in animal blood, brain (0.14%), muscle (0.50%), and testes (0.18%). The liver and kidney contain approximately 0.01% creatine.
Creatine was identified in 1832 when Michel Eugène Chevreul isolated it from the basified water-extract of skeletal muscle. He later named the crystallized precipitate after the Greek word for meat, κρέας (kreas).
Apart from its pharmacological effects, creatine content (as a percentage of crude protein) can be used as an indicator of meat quality.[3]
In solution, creatine is in equilibrium with creatinine.[4] Creatine is a derivative of the guanidinium cation.
Biosynthesis
Creatine is not an essential nutrient[5] as it is naturally produced in the human body from the amino acids glycine and arginine. In the first step of the biosynthesis these two amino acids are combined by the enzyme arginine:glycine amidinotransferase (AGAT, EC:2.1.4.1) to form guanidinoacetate, which is then methylated by guanidinoacetate N-methyltransferase (GAMT,EC:2.1.1.2), using S-adenosyl methionine as the methyl donor. Creatine itself can be phosphorylated by creatine kinase to form phosphocreatine, which is used as an energy buffer in skeletal muscles and the brain.
.png)
Synthesis primarily takes place in the kidney and liver, with creatine then being transported to the muscles via the blood. Approximately 95% of the human body's total creatine is located in skeletal muscle.[6] In humans and animals, approximately half of stored creatine originates from food (about 1 g/day, mainly from meat).[5] Some small studies suggest that total muscle creatine is significantly lower in vegetarians than non-vegetarians, as expected since vegetables are not a primary source of creatine. However, subjects happened to show the same levels after using supplements.[7] Given that creatine can be synthesized from the above-mentioned amino acids, protein sources rich in these amino acids can be expected to provide sufficient native biosynthesis in the body.[5]
Genetic deficiencies in the creatine biosynthetic pathway lead to various severe neurological defects.[8] Clinically, there are three distinct disorders of creatine metabolism. Deficiencies in the two synthesis enzymes can cause L-arginine:glycine amidinotransferase deficiency and guanidinoacetate methyltransferase deficiency. Both biosynthetic defects are inherited in an autosomal recessive manner. A third defect, creatine transporter defect is caused by mutations in SLC6A8 and inherited in a X-linked manner. This condition is related to the transport of creatine into the brain.[9]
Phosphocreatine system
Creatine, synthesized in the liver and kidney, is transported through the blood and taken up by tissues with high energy demands, such as the brain and skeletal muscle, through an active transport system. The concentration of ATP in skeletal muscle is usually 2-5 mM, which would result in a muscle contraction of only a few seconds.[10] Fortunately, during times of increased energy demands, the phosphagen (or ATP/PCr) system rapidly resynthesizes ATP from ADP with the use of phosphocreatine (PCr) through a reversible reaction with the enzyme creatine kinase (CK). In skeletal muscle, PCr concentrations may reach 20-35 mM or more. Additionally, in most muscles, the ATP regeneration capacity of CK is very high and is therefore not a limiting factor. Although the cellular concentrations of ATP are small, changes are difficult to detect because ATP is continuously and efficiently replenished from the large pools of PCr and CK.[10] Creatine has the ability to increase muscle stores of PCr, potentially increasing the muscle’s ability to resynthesize ATP from ADP to meet increased energy demands.[11] [12][13]

Arg - Arginine; GATM - Glycine amidinotransferase; GAMT - Guanidinoacetate N-methyltransferase; Gly - Glycine; Met - Methionine; SAH - S-adenosyl homocysteine; SAM - S-adenosyl methionine. ---- The color scheme is as follows:enzymes, coenzymes and the Met part, substrate names, the Gly part, the Arg part
Health effects
Supplements
Creatine supplements are used by athletes, bodybuilders, wrestlers, sprinters, and others who wish to gain muscle mass. The Mayo Clinic states that creatine has been associated with asthmatic symptoms and warns against consumption by persons with known allergies to creatine.[14]
A 2009 systematic review discredited concerns that creatine supplementation could affect hydration status and heat tolerance and lead to muscle cramping and diarrhea.[15][16]
There are reports of kidney damage with creatine use, such as interstitial nephritis; patients with kidney disease should avoid use of this supplement.[14] In similar manner, liver function may be altered, and caution is advised in those with underlying liver disease, although studies have shown little or no adverse impact on kidney or liver function from oral creatine supplementation.[17] In 2004 the European Food Safety Authority (EFSA) published a record which stated that oral long-term intake of 3 g pure creatine per day is risk-free.[18] A 2003 study on athletes who took creatine for 21 months found no significant changes in markers of renal function;[19] a 2008 study on athletes who took creatine for 3 months found no evidence of kidney damage during that time.[20]
Long-term administration of large quantities of creatine is reported to increase the production of formaldehyde, which has the potential to cause serious unwanted side effects. However, this risk is largely theoretical because urinary excretion of formaldehyde, even under heavy creatine supplementation, does not exceed normal limits.[21][22]
Extensive research has shown that oral creatine supplementation at a rate of five to 20 grams per day appears to be very safe and largely devoid of adverse side-effects,[23] while at the same time effectively improving the physiological response to resistance exercise, increasing the maximal force production of muscles in both men and women.[24][25]
Many meta analyses found that creatine treatment resulted in no abnormal renal, hepatic, cardiac, or muscle function.[26][27] It is ineffective as a treatment for amyotrophic lateral sclerosis.[28]
While some research indicates that supplementation with pure creatine is safe, a survey of 33 commercially available in Italy supplements found that over 50% of them exceeded the European Food Safety Authority recommendations in at least one contaminant. The most prevalent of these contaminants was creatinine, a breakdown product of creatine also produced by the body.[29] Creatinine was present in higher concentrations than the European Food Safety Authority recommendations in 44% of the samples. About 15% of the samples had detectable levels of dihydro-1,3,5-triazine or a high dicyandiamide concentration. Heavy metals contamination was not found to be a concern, with only minor levels of mercury being detectable. Two studies reviewed in 2007 found no impurities.[26]
Pharmacokinetics
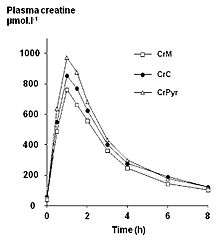
Endogenous serum or plasma creatine concentrations in healthy adults are normally in a range of 2–12 mg/L. A single 5 g (5000 mg) oral dose in healthy adults results in a peak plasma creatine level of approximately 120 mg/L at 1–2 hours post-ingestion. Creatine has a fairly short elimination half-life, averaging just less than 3 hours, so to maintain an elevated plasma level it would be necessary to take small oral doses every 3–6 hours throughout the day. After the "loading dose" period (1–2 weeks, 12-24 g a day), it is no longer necessary to maintain a consistently high serum level of creatine. As with most supplements, each person has their own genetic "preset" amount of creatine they can hold. The rest is eliminated as waste. A typical post-loading dose is 2-5 g daily.[31][32][33]
Pregnancy and breastfeeding
While research is limited, some recent retrospective studies show higher maternal serum creatine to correlate with higher birth weight and better birth outcomes. However, as of 2014, there were no randomised controlled trials establishing its effectiveness when supplemented, for neuroprotection of the fetus.
Pasteurized cow's milk contains higher levels of creatine than human milk.[34][35]
Muscle disorders
A meta analysis found that creatine treatment increased muscle strength in muscular dystrophies, and potentially improved functional performance.[36] It has also been implicated in decreasing mutagenesis in DNA.[37]
Heterocyclic amines
When creatine is mixed with protein and sugar at high temperatures (above 148 °C), the resulting reaction produces heterocyclic amines (HCAs). Such a reaction happens when grilling or pan frying meat.[38]
A Meta-analysis from 2011 concluded that "search for the excretion of heterocyclic amines remains a future task to definitively exclude the unproved allegation made by some national agencies".[27]
See also
References
- ↑ Entry "creatine" in Merriam-Webster Online Dictionary.
- ↑ Wells, J. C. (2000). Longman Pronunciation Dictionary. Harlow, England: Pearson Education Ltd.
- ↑ http://pubs.acs.org/doi/abs/10.1021/jf60128a026?journalCode=jafcau
- ↑ Cannan, R. K.; Shore, A. (1928). "The creatine-creatinine equilibrium. The apparent dissociation constants of creatine and creatinine" (PDF). Biochem. J. 22 (4): 920–29. PMC 1252207
 . PMID 16744118. Retrieved 2010-10-29.
. PMID 16744118. Retrieved 2010-10-29. - 1 2 3 "Creatine". Beth Israel Deaconess Medical Center. Retrieved 2010-08-23.
- ↑ "Creatine". MedLine Plus Supplements. U.S. National Library of Medicine. 20 July 2010. Retrieved 2010-08-16.
- ↑ Burke DG, Chilibeck PD, Parise G, Candow DG, Mahoney D, Tarnopolsky M; Chilibeck; Parise; Candow; Mahoney; Tarnopolsky (2003). "Effect of creatine and weight training on muscle creatine and performance in vegetarians". Medicine and science in sports and exercise. 35 (11): 1946–55. doi:10.1249/01.MSS.0000093614.17517.79. PMID 14600563.
- ↑ "L-Arginine:Glycine Amidinotransferase". Retrieved 2010-08-16.
- ↑ Braissant, O; Henry, H; Béard, E; Uldry, J (May 2011). "Creatine deficiency syndromes and the importance of creatine synthesis in the brain.". Amino Acids. 40 (5): 1315–24. doi:10.1007/s00726-011-0852-z. PMID 21390529.
- 1 2 Wallimann, T; Wyss, M; Brdiczka, D; Nicolay, K; Eppenberger, HM (January 1992). "Intracellular compartmentation, structure and function of creatine kinase isoenzymes in tissues with high and fluctuating energy demands: the 'phosphocreatine circuit' for cellular energy homeostasis". The Biochemical Journal. 281 (Pt 1): 21–40. doi:10.1042/bj2810021. PMC 1130636
 . PMID 1731757.
. PMID 1731757. - ↑ Spillane, M; Schoch, R; Cooke, M; Harvey, T; Greenwood, M; Kreider, R; Willoughby, DS (2009). "The effects of creatine ethyl ester supplementation combined with heavy resistance training on body composition, muscle performance, and serum and muscle creatine levels". Journal of the International Society of Sports Nutrition. 6 (1): 6. doi:10.1186/1550-2783-6-6. PMC 2649889
 . PMID 19228401.
. PMID 19228401. - ↑ "The creatine kinase system and pleiotropic effects of creatine". Amino Acids. 40 (5): 1271–96. May 2011. doi:10.1007/s00726-011-0877-3. PMC 3080659
 . PMID 21448658..
. PMID 21448658.. - ↑ T. Wallimann, M. Tokarska-Schlattner, D. Neumann u. a.: The Phosphocreatine Circuit: Molecular and Cellular Physiology of Creatine Kinases, Sensitivity to Free Radicals, and Enhancement by Creatine Supplementation. In: Molecular System Bioenergetics: Energy for Life. 22. November 2007. doi:10.1002/9783527621095.ch7C
- 1 2 "Creatine: Safety". MayoClinic.com. Retrieved 2010-08-16.
- ↑ Lopez RM, Casa DJ, McDermott BP, Ganio MS, Armstrong LE, Maresh CM; Casa; McDermott; Ganio; Armstrong; Maresh (2009). "Does Creatine Supplementation Hinder Exercise Heat Tolerance or Hydration Status? A Systematic Review With Meta-Analyses". Journal of Athletic Training. 44 (2): 215–23. doi:10.4085/1062-6050-44.2.215. PMC 2657025
 . PMID 19295968.
. PMID 19295968. - ↑ Dalbo VJ, Roberts MD, Stout JR, Kerksick CM; Roberts; Stout; Kerksick (July 2008). "Putting to rest the myth of creatine supplementation leading to muscle cramps and dehydration". British Journal of Sports Medicine. 42 (7): 567–73. doi:10.1136/bjsm.2007.042473. PMID 18184753.
- ↑ Poortmans JR, Francaux M; Francaux (September 2000). "Adverse effects of creatine supplementation: fact or fiction?". Sports Medicine. 30 (3): 155–70. doi:10.2165/00007256-200030030-00002. PMID 10999421.
- ↑ http://www.efsa.europa.eu/EFSA/efsa_locale-1178620753824_1178620761727.htm
- ↑ Kreider, Richard B.; Melton, Charles; Rasmussen, Christopher J.; Greenwood, Michael; Lancaster, Stacy; Cantler, Edward C.; Milnor, Pervis; Almada, Anthony L. (2003). "Long-term creatine supplementation does not significantly affect clinical markers of health in athletes". Molecular and Cellular Biochemistry. 244 (1–2): 95–104. doi:10.1023/A:1022469320296. PMID 12701816.
- ↑ Gualano, Bruno; Ugrinowitsch, Carlos; Novaes, Rafael Batista; Artioli, Guilherme Gianini; Shimizu, Maria Heloisa; Seguro, Antonio Carlos; Harris, Roger Charles; Lancha, Antonio Herbert (2008). "Effects of creatine supplementation on renal function: A randomized, double-blind, placebo-controlled clinical trial". European Journal of Applied Physiology. 103 (1): 33–40. doi:10.1007/s00421-007-0669-3. PMID 18188581.
- ↑ Francaux M, Poortmans JR; Poortmans (December 2006). "Side effects of creatine supplementation in athletes". International Journal of Sports Physiology and Performance. 1 (4): 311–23. PMID 19124889.
- ↑ "International Society of Sports Nutrition position stand: creatine supplementation and exercise". jissn. Retrieved 19 January 2012.
- ↑ Bizzarini E, De Angelis L; De Angelis (December 2004). "Is the use of oral creatine supplementation safe?". The Journal of Sports Medicine and Physical Fitness. 44 (4): 411–6. PMID 15758854.
- ↑ Bemben MG, Lamont HS; Lamont (2005). "Creatine supplementation and exercise performance: recent findings". Sports Medicine. 35 (2): 107–25. doi:10.2165/00007256-200535020-00002. PMID 15707376.
- ↑ Kreider RB (February 2003). "Effects of creatine supplementation on performance and training adaptations" (PDF). Molecular and Cellular Biochemistry. 244 (1–2): 89–94. doi:10.1023/A:1022465203458. PMID 12701815.
- 1 2 Persky, AM; Rawson, ES (2007). "Safety of creatine supplementation.". Sub-cellular biochemistry. 46: 275–89. doi:10.1007/978-1-4020-6486-9_14. PMID 18652082.
- 1 2 Kim HJ, Kim CK, Carpentier A, Poortmans JR (2011). "Studies on the safety of creatine supplementation". Amino Acids (Review). 40 (5): 1409–18. doi:10.1007/s00726-011-0878-2. PMID 21399917.
- ↑ Pastula, DM; Moore, DH; Bedlack, RS (12 December 2012). "Creatine for amyotrophic lateral sclerosis/motor neuron disease.". The Cochrane database of systematic reviews. 12: CD005225. PMID 23235621.
- ↑ Moreta S, Prevarin A, Tubaro F.; Prevarin; Tubaro (June 2011). "Levels of creatine, organic contaminants and heavy metals in creatine dietary supplements". Food Chemistry. 126 (3): 1232–1238. doi:10.1016/j.foodchem.2010.12.028.
- ↑ Jäger R, Harris RC, Purpura M, Francaux M (2007). "Comparison of new forms of creatine in raising plasma creatine levels". J Int Soc Sports Nutr. 4: 17. doi:10.1186/1550-2783-4-17. PMC 2206055
 . PMID 17997838.
. PMID 17997838. - ↑ Kamber M, Koster M, Kreis R, Walker G, Boesch C, Hoppeler H. Creatine supplementation--part I: performance, clinical chemistry, and muscle volume. Med. Sci. Sports Exer. 31: 1763-1769, 1999.
- ↑ Deldicque, L; Décombaz, J; Zbinden Foncea, H; Vuichoud, J; Poortmans, JR; Francaux, M (2008). "Kinetics of creatine ingested as a food ingredient". Eur. J. Appl. Physiol. 102: 133–143. doi:10.1007/s00421-007-0558-9. PMID 17851680.
- ↑ R. Baselt, Disposition of Toxic Drugs and Chemicals in Man, 8th edition, Biomedical Publications, Foster City, CA, 2008, pp. 366-368.
- ↑ Hülsemann J, Manz F, Wember T, Schöch G; Manz; Wember; Schöch (1987). "[Administration of creatine and creatinine with breast milk and infant milk preparations]". Klinische Pädiatrie (in German). 199 (4): 292–5. doi:10.1055/s-2008-1026805. PMID 3657037.
- ↑ Wallimann, Theo; Tokarska-Schlattner, Malgorzata; Schlattner, Uwe (1 May 2011). "The creatine kinase system and pleiotropic effects of creatine". Amino Acids. Springer Wien. 40 (5): 1271–1296. doi:10.1007/s00726-011-0877-3. ISSN 0939-4451. PMC 3080659
 . PMID 21448658.
. PMID 21448658. - ↑ Kley, R. A.; Tarnopolsky, M. A.; Vorgerd, M. (2011). Kley, Rudolf A, ed. "Creatine for treating muscle disorders". The Cochrane Library (2): CD004760. doi:10.1002/14651858.CD004760.pub3. PMID 21328269.
- ↑ Rahimi, R. (2011). "Creatine Supplementation Decreases Oxidative DNA Damage and Lipid Peroxidation Induced by a Single Bout of Resistance Exercise". Journal of Strength and Conditioning Research. 25 (12): 3448–3455. doi:10.1519/JSC.0b013e3182162f2b. PMID 22080314.
- ↑ "Chemicals in Meat Cooked at High Temperatures and Cancer Risk". National Cancer Institute.
External links
- Creatine bound to proteins in the PDB
- International Society of Sports Nutrition position stand: Creatine