Estrogen insensitivity syndrome
| Estrogen insensitivity syndrome | |
|---|---|
 | |
| EIS results when the function of the estrogen receptor alpha (ERα) is impaired. The ERα protein (pictured) mediates most of the effects of estrogens in the human body. | |
| Classification and external resources | |
| Specialty | Endocrinology |
| OMIM | 133430 |
Estrogen insensitivity syndrome (EIS), or estrogen resistance, is a form of congenital estrogen deficiency[1] caused by a defective estrogen receptor (ER), specifically ERα. Thus, estrogens cannot be recognized and initiate their biological action.[2] Congenital estrogen deficiency can also be caused by a defect in the aromatase enzyme.[3]
In humans, the condition is very rare and only two cases have been described, one male and one female.[4][5]
Case reports
Male case
In 1994, a 28-year-old man with EIS was reported.[6][7] He was fully masculinized.[8] At 204 cm, he had tall stature.[6] His epiphyses were unfused, and there was evidence of still-occurring slow linear growth (for comparison, his height at 16 years of age was 178 cm).[6] He also had markedly delayed skeletal maturation (bone age 15 years), a severely undermineralized skeleton, evidence of increased bone resorption, and very early-onset osteoporosis.[6] The genitalia, testes, and prostate of the patient were all normal and of normal size/volume.[6] The sperm count of the patient was normal (25 million/mL; normal, >20 million/mL), but his sperm viability was low (18%; normal, >50%), indicating some degree of infertility.[6] The patient also had early-onset temporal hair loss.[6] He reported no history of gender identity disorder, considered himself to have strong heterosexual interests, and had normal sexual function, including morning erections and nocturnal emissions.[6]
Follicle-stimulating hormone and luteinizing hormone levels were considerably elevated (30–33 mIU/mL and 34–37 mIU/mL, respectively) and estradiol and estrone levels were markedly elevated (145 pg/mL and 119–272 pg/mL, respectively), while testosterone levels were normal (445 ng/dL).[6] Sex hormone-binding globulin levels were mildly elevated (6.0–10.0 nmol/L), while thyroxine-binding globulin, corticosteroid-binding globulin, and prolactin levels were all normal.[6] Osteocalcin and bone-specific alkaline phosphatase levels were both substantially elevated (18.7–21.6 ng/mL and 33.3–35.9 ng/mL, respectively).[6]
Treatment with up to extremely high doses of ethinyl estradiol (fourteen 100-µg patches per week) had no effect on any of his symptoms of hypoestrogenism, did not produce any estrogenic effects such as gynecomastia, and had no effect on any of his physiological parameters (e.g., hormone levels or bone parameters), suggesting a profile of complete estrogen insensitivity syndrome.[6]
Female case
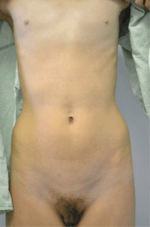
In 2013, an 18-year-old woman with EIS was reported.[4][5] DNA sequencing revealed a homozygous mutation in ESR1, the gene that encodes the ERα.[5] Within the ligand-binding domain, the neutral polar glutamine 375 was changed to a basic, polar histidine.[5] An in vitro assay of ERα-dependent gene transcription found that the EC50 for transactivation had been reduced by 240-fold relative to normal, non-mutated ERα, indicating an extreme reduction in the activity of the receptor.[5] Clinical signs suggested a profile of complete estrogen insensitivity syndrome with a resemblance to ERα knockout mice.[5] The patient presented with delayed puberty, including an absence of breast development (Tanner stage I) and primary amenorrhea, as well as intermittent pelvic pain.[5] Examination revealed markedly enlarged ovaries with multiple hemorrhagic cysts as the cause of the lower abdominal pain.[5]
Estrogen levels were dramatically and persistently elevated (estradiol levels were 2340 pg/mL, regarded as being about 10 times the normal level, and ranged from 750–3500 pg/mL), gonadotropin levels were mildly elevated (follicle-stimulating hormone and luteinizing hormone levels were 6.7–19.1 mIU/mL and 5.8–13.2 mIU/mL, respectively), and testosterone levels were slightly elevated (33–88 ng/dL).[5] Inhibin A levels were also markedly elevated.[5] Sex hormone-binding globulin, corticosteroid-binding globulin, thyroxine-binding globulin, prolactin, and triglycerides, which are known to be elevated by estrogen, were all within normal ranges in spite of the extremely high levels of estrogen, and inhibin B levels were also normal.[5] Her relatively mildly elevated levels of gonadotropins were attributed to retained negative feedback by progesterone as well as by her elevated levels of testosterone and inhibin A, although it was acknowledged that possible effects of estrogen mediated by other receptors such as ERβ could not be excluded.[5]
The patient had a small uterus, with an endometrial stripe that could not be clearly identified.[5] At the age of 15 years, 5 months, her bone age was 11 or 12 years, and at the age of 17 years, 8 months, her bone age was 13.5 years.[5] Her bone mass was lower than expected for her age, and levels of osteocalcin and C-terminal telopeptide were both elevated, suggesting an increased rate of bone turnover.[5] She was 162.6 cm tall, and her growth velocity indicated a lack of estrogen-induced growth spurt at puberty.[5] The patient had normal pubic hair development (Tanner stage IV) and severe facial acne, which could both be attributed to testosterone.[5] Her ovarian pathology was attributed to the elevated levels of gonadotropins.[5] In addition to her absence of breast development and areolar enlargement, the patient also appeared to show minimal widening of the hips and a lack of subcutaneous fat deposition, which is in accordance with the established role of estrogen and ERα in the development of female secondary sexual characteristics.[5][9]
Treatment of the patient with conjugated equine estrogens and high doses of estradiol had no effect.[5] Although the authors of the paper considered her ERα to be essentially unresponsive to estrogen, they stated that they "[could not] exclude the possibility that some residual estrogen sensitivity could be present in some tissues", which is in accordance with the fact that the EC50 of her ERα had been reduced 240-fold but had not been abolished.[5] Treatment with a progestin, norethisterone, reduced her estradiol concentrations to normal levels and decreased the size of her ovaries and the number of ovarian cysts, alleviating her hypothalamic-pituitary-gonadal axis hyperactivity and ovarian pathology.[5]
ER knockout mice
EIS can be experimentally induced in animals via knockout of the ER. In so-called ERKO mice, different ERs can be disabled allowing to study the role of such receptors.[10] ERKO mice show development of the respective female or male reproductive systems, and male and female α-ERKO mice are infertile, β-ERKO males are fertile while females are subfertile, male and female double α- and β-ERKO mice are sterile. The uterus and mammary glands are hypoplastic and do not respond to exogenous stimulation by estrogens. Males are infertile with atrophy in the testes. Bones age is delayed and bones are more brittle. Variations in these patterns can be achieved by selectively disabling the α or β ERs.
Androgen insensitivity syndrome
In contrast to EIS, androgen insensitivity syndrome (AIS), a condition in which the androgen receptor is defective, is relatively common. This can be explained by the genetics of each syndrome. AIS is a X-linked recessive condition and thus carried over, by females, into future generations (although the most severe form, complete androgen insensitivity syndrome (CAIS), results in sterility, and hence cannot be passed on to offspring). EIS is not compatible with reproduction, thus each occurrence in humans would have to be a de novo mutation and is not transmitted to offspring.
See also
References
- ↑ Rochira V, Balestrieri A, Madeo B, Baraldi E, Faustini-Fustini M, Granata AR, Carani C, et al. (June 2001). "Congenital estrogen deficiency: in search of the estrogen role in human male reproduction". Molecular and Cellular Endocrinology. 178 (1-2): 107–15. doi:10.1016/S0303-7207(01)00432-4. PMID 11403900.
- ↑ Smith EP, Boyd J, Frank GR, Takahashi H, Cohen RM, Specker B, Williams TC, Lubahn DB, Korach KS, et al. (October 1994). "Estrogen resistance caused by a mutation in the estrogen-receptor gene in a man". The New England Journal of Medicine. 331 (16): 1056–61. doi:10.1056/NEJM199410203311604. PMID 8090165.
- ↑ Rochira V, Balestrieri A, Madeo B, Spaggiari A, Carani C (July 2002). "Congenital estrogen deficiency in men: a new syndrome with different phenotypes; clinical and therapeutic implications in men". Molecular and Cellular Endocrinology. 193 (1-2): 19–28. doi:10.1016/S0303-7207(02)00092-8. PMID 12160998.
- 1 2 J. Larry Jameson; Leslie J. De Groot (25 February 2015). Endocrinology: Adult and Pediatric. Elsevier Health Sciences. pp. 238–. ISBN 978-0-323-32195-2.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 Quaynor SD, Stradtman EW, Kim HG, Shen Y, Chorich LP, Schreihofer DA, Layman LC (July 2013). "Delayed puberty and estrogen resistance in a woman with estrogen receptor α variant". The New England Journal of Medicine. 369 (2): 164–71. doi:10.1056/NEJMoa1303611. PMC 3823379
 . PMID 23841731.
. PMID 23841731. - 1 2 3 4 5 6 7 8 9 10 11 12 Smith EP, Boyd J, Frank GR, Takahashi H, Cohen RM, Specker B, Williams TC, Lubahn DB, Korach KS (October 1994). "Estrogen resistance caused by a mutation in the estrogen-receptor gene in a man". The New England Journal of Medicine. 331 (16): 1056–61. doi:10.1056/NEJM199410203311604. PMID 8090165.
- ↑ Korach KS, Couse JF, Curtis SW, Washburn TF, Lindzey J, Kimbro KS, Eddy EM, Migliaccio S, Snedeker SM, Lubahn DB, Schomberg DW, Smith EP (1996). "Estrogen receptor gene disruption: molecular characterization and experimental and clinical phenotypes". Recent Progress in Hormone Research. 51: 159–86; discussion 186–8. PMID 8701078.
- ↑ Gene Therapy. Academic Press. 12 August 1997. pp. 344–. ISBN 978-0-08-058132-3.
- ↑ Thomas L. Lemke; David A. Williams (24 January 2012). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. pp. 1392–. ISBN 978-1-60913-345-0.
- ↑ Couse JF, Korach KS (June 1999). "Estrogen receptor null mice: what have we learned and where will they lead us?". Endocrine Reviews. 20 (3): 358–417. doi:10.1210/er.20.3.358. PMID 10368776.