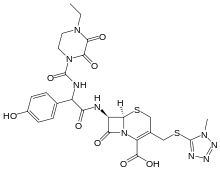
Cefoperazone
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a601206 |
| ATC code | J01DD12 (WHO) QJ51DD12 (WHO) |
| Pharmacokinetic data | |
| Excretion | Hepatic |
| Identifiers | |
| |
| CAS Number |
62893-19-0 |
| PubChem (CID) | 44185 |
| DrugBank |
DB01329 |
| ChemSpider |
40206 |
| UNII |
7U75I1278D |
| KEGG |
D07645 |
| ChEMBL |
CHEMBL507674 |
| ECHA InfoCard | 100.057.936 |
| Chemical and physical data | |
| Formula | C25H27N9O8S2 |
| Molar mass | 645.67 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Cefoperazone is a third-generation cephalosporin antibiotic, marketed by Pfizer under the name Cefobid. It is one of few cephalosporin antibiotics effective in treating Pseudomonas bacterial infections which are otherwise resistant to these antibiotics.
Cefoperazone is also sold as a co-formulation with sulbactam. Cefoperazone exerts its bactericidal effect by inhibiting the bacterial cell wall synthesis, and sulbactam acts as a beta-lactamase inhibitor, to increase the antibacterial activity of cefoperazone against beta-lactamase-producing organisms.
Spectrum of bacterial susceptibility
Cefoperazone has a broad spectrum of activity and has been used to target bacteria responsible for causing infections of the respiratory and urinary tract, skin, and the female genital tract. The following represents MIC susceptibility data for a few medically significant microorganisms.
- Haemophilus influenzae: 0.12 - 0.25 µg/ml
- Staphylococcus aureus: 0.125 - 32 µg/ml
- Streptococcus pneumoniae: ≤0.007 - 1 µg/ml
Adverse effects
Cefoperazone contains an N-methylthiotetrazole (NMTT or 1-MTT) side chain. As the antibiotic is broken down in the body, it releases free NMTT, which can cause hypoprothrombinemia (likely due to inhibition of the enzyme vitamin K epoxide reductase) and a reaction with ethanol similar to that produced by disulfiram (Antabuse), due to inhibition of aldehyde dehydrogenase.[3]
References
- ↑ http://antibiotics.toku-e.com/antimicrobial_462_1.html
- ↑ http://www.toku-e.com/Assets/MIC/Cefoperazone%20sodium.pdf
- ↑ Stork CM (2006). "Antibiotics, antifungals, and antivirals". In Nelson LH, Flomenbaum N, Goldfrank LR, Hoffman RL, Howland MD, Lewin NA. Goldfrank's toxicologic emergencies. New York: McGraw-Hill. p. 847. ISBN 0-07-143763-0. Retrieved 2009-07-03.