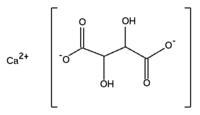
Calcium tartrate
 | |
| Names | |
|---|---|
| IUPAC name
2,3-Dihydroxybutanedioic acid calcium salt | |
| Identifiers | |
| 3164-34-9 (anhydrous) 5892-21-7 (tetrahydrate) | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 10606089 |
| ECHA InfoCard | 100.019.656 |
| E number | E354 (antioxidants, ...) |
| PubChem | 3083790 |
| |
| |
| Properties | |
| CaC4H4O6 | |
| Molar mass | 190.16484 g/mol (anhydrous) 260.21 g/mol (tetrahydrate) |
| Appearance | hygroscopic white powder or colorless crystals |
| Density | 1.817 g/cm3 (tetrahydrate) |
| Melting point | tetrahydrate decomposes at 160 °C anhydrous decomposes at 650 °C |
| 0.037 g/100 ml (0 °C) 0.2 g/100 ml (85 °C) | |
| Structure | |
| d or l rhombic dl triclinic | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Calcium tartrate is a byproduct of the wine industry, prepared from wine fermentation dregs. It is the calcium salt of tartaric acid, an acid most commonly found in grapes. Its solubility decreases with lower temperature, which results in the forming of whitish (in red wine often reddish) crystalline clusters as it precipitates. It finds use as a food preservative and acidity regulator. Like tartaric acid, calcium tartrate has two asymmetric carbons, hence it has two chiral isomers and a non-chiral isomer (meso-form). Most calcium tartrate of biological origin is the chiral levorotatory (–) isomer.
References
This article is issued from Wikipedia - version of the 5/27/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.