Calcite sea
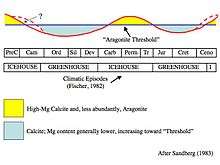
A calcite sea is one in which low-magnesium calcite is the primary inorganic marine calcium carbonate precipitate. An aragonite sea is the alternate seawater chemistry in which aragonite and high-magnesium calcite are the primary inorganic carbonate precipitates. The Early Paleozoic and the Middle to Late Mesozoic oceans were predominantly calcite seas, whereas the Middle Paleozoic through the Early Mesozoic and the Cenozoic (including today) are characterized by aragonite seas (Wilkinson et al., 1985; Wilkinson and Given, 1986; Morse and Mackenzie, 1990; Lowenstein et al., 2001; Palmer and Wilson, 2004).

The most significant geological and biological effects of calcite sea conditions include rapid and widespread formation of carbonate hardgrounds (Palmer, 1982; Palmer et al., 1988; Wilson and Palmer, 1992), calcitic ooids (Sandberg, 1983; Wilkinson et al., 1985), calcite cements (Wilkinson and Given, 1986), and the contemporaneous dissolution of aragonite shells in shallow warm seas (Cherns and Wright, 2000; Palmer and Wilson, 2004). Hardgrounds were very common, for example, in the calcite seas of the Ordovician and Jurassic, but virtually absent from the aragonite seas of the Permian (Palmer, 1982).
Fossils of invertebrate organisms found in calcite sea deposits are usually dominated by either thick calcite shells and skeletons (Wilkinson, 1979; Stanley and Hardie, 1998, 1999; Porter, 2007), were infaunal and/or had thick periostraca (Pojeta, 1971), or had an inner shell of aragonite and an outer shell of calcite (Harper et al., 1997). This was apparently because aragonite dissolved quickly on the seafloor and had to be either avoided or protected as a biomineral (Palmer and Wilson, 2004).
Calcite seas were coincident with times of rapid seafloor spreading and global greenhouse climate conditions (Stanley and Hardie, 1999). Seafloor spreading centers cycle seawater through hydrothermal vents, reducing the ratio of magnesium to calcium in the seawater through metamorphism of calcium-rich minerals in basalt to magnesium-rich clays (Wilkinson and Given, 1986; Lowenstein et al., 2001). This reduction in the Mg/Ca ratio favors the precipitation of calcite over aragonite. Increased seafloor spreading also means increased volcanism and elevated levels of carbon dioxide in the atmosphere and oceans. This may also have an effect on which polymorph of calcium carbonate is precipitated (Lowenstein et al., 2001). Further, high calcium concentrations of seawater favor the burial of CaCO3, thereby removing alkalinity from the ocean, lowering seawater pH and reducing its acid/base buffering.[1]
-

Table showing the conditions for calcite and aragonite seas.
-

Tectonic mechanism for changing Mg/Ca ratios in seawater.
-

The Ordovician cystoid Echinosphaerites filled with early calcite cements.
-

Cartoon demonstrating the growth of internal calcite cements and external calcite cements associated with the Ordovician cystoid Echinosphaerites.
-

An encrusted Ordovician bivalve external mold showing contemporaneous dissolution of the original aragonite shell and calcitic cementation of the mold.
-

An encrusted Ordovician nautiloid internal mold showing contemporaneous dissolution of the original aragonite shell and calcitic cementation.
-

The boring Palaeosabella in an Ordovician bivalve shell. The borings penetrated an inner aragonitic shell layer which dissolved away.
-

Petroxestes borings in an Upper Ordovician hardground, southern Ohio.
References
- ↑ Hain, Mathis P.; Sigman, Daniel M.; Higgins, John A.; Haug, Gerald H. (2015). "The effects of secular calcium and magnesium concentration changes on the thermodynamics of seawater acid/base chemistry: Implications for Eocene and Cretaceous ocean carbon chemistry and buffering" (PDF). Global Biogeochemical Cycles. 29 (5): 517–533. Bibcode:2015GBioC..29..517H. doi:10.1002/2014GB004986. ISSN 0886-6236.
- Cherns, L.; Wright, V.P. (2000). "Missing molluscs as evidence of large-scale, early skeletal aragonite dissolution in a Silurian Sea". Geology. 28 (9): 791–794. Bibcode:2000Geo....28..791C. doi:10.1130/0091-7613(2000)28<791:MMAEOL>2.0.CO;2.
- Harper, E.M.; Palmer, T.J.; Alphey, J.R. (1997). "Evolutionary response by bivalves to changing Phanerozoic sea-water chemistry". Geological Magazine. 134 (3): 403–407. doi:10.1017/S0016756897007061.
- Lowenstein, T.K.; Timofeeff, M.N.; Brennan, S.T.; Hardie, L.A.; Demicco, R.V. (2001). "Oscillations in Phanerozoic seawater chemistry: evidence from fluid inclusions". Science. 294 (5544): 1086–1088. Bibcode:2001Sci...294.1086L. doi:10.1126/science.1064280. PMID 11691988.
- Morse, J.W.; Mackenzie, F.T. (1990). "Geochemistry of sedimentary carbonates". Developments in Sedimentology. 48: 1–707. doi:10.1016/S0070-4571(08)70330-3.
- Palmer, T.J.; Wilson, M.A. (2004). "Calcite precipitation and dissolution of biogenic aragonite in shallow Ordovician calcite seas". Lethaia. 37 (4): 417–427 . doi:10.1080/00241160410002135.
- Palmer, T.J. (1982). "Cambrian to Cretaceous changes in hardground communities". Lethaia. 15 (4): 309–323. doi:10.1111/j.1502-3931.1982.tb01696.x.
- Palmer, T.J.; Hudson, J.D.; Wilson, M.A. (1988). "Palaeoecological evidence for early aragonite dissolution in ancient calcite seas". Nature. 335 (6193): 809–810. Bibcode:1988Natur.335..809P. doi:10.1038/335809a0.
- Pojeta, J. Jr. (1988). "Review of Ordovician pelecypods". U.S. Geological Survey Professional Paper. 1044: 1–46.
- Porter, S.M. (2007). "Seawater chemistry and early carbonate biomineralization". Science. 316 (5829): 1302–1304. Bibcode:2007Sci...316.1302P. doi:10.1126/science.1137284. PMID 17540895.
- Sandberg, P.A. (1983). "An oscillating trend in Phanerozoic non-skeletal carbonate mineralogy". Nature. 305 (5929): 19–22. Bibcode:1983Natur.305...19S. doi:10.1038/305019a0.
- Stanley, S.M.; Hardie, L.A. (1998). "Secular oscillations in the carbonate mineralogy of reef-building and sediment-producing organisms driven by tectonically forced shifts in seawater chemistry". Palaeogeography, Palaeoclimatology, Palaeoecology. 144 (1-2): 3–19. doi:10.1016/S0031-0182(98)00109-6.
- Stanley, S.M.; Hardie, L.A. (1999). "Hypercalcification; paleontology links plate tectonics and geochemistry to sedimentology". GSA Today. 9: 1–7.
- Wilkinson, B.H. (1979). "Biomineralization, paleooceanography, and the evolution of calcareous marine organisms". Geology. 7 (11): 524–527. Bibcode:1979Geo.....7..524W. doi:10.1130/0091-7613(1979)7<524:BPATEO>2.0.CO;2.
- Wilkinson, B.H.; Given, K.R. (1986). "Secular variation in abiotic marine carbonates: constraints on Phanerozoic atmospheric carbon dioxide contents and oceanic Mg/Ca ratios". Journal of Geology. 94 (3): 321–333. Bibcode:1986JG.....94..321W. doi:10.1086/629032.
- Wilkinson, B.H.; Owen, R.M.; Carroll, A.R. (1985). "Submarine hydrothermal weathering, global eustacy, and carbonate polymorphism in Phanerozoic marine oolites". Journal of Sedimentary Petrology. 55: 171–183.
- Wilson, M.A.; Palmer, T.J. (1992). "Hardgrounds and hardground faunas". University of Wales, Aberystwyth, Institute of Earth Studies Publications. 9: 1–131.