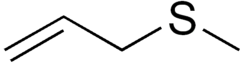
Allyl methyl sulfide
 | |
| Names | |
|---|---|
| Preferred IUPAC name
3-Methylsulfanylprop-1-ene | |
| Other names
Methyl propenyl sulfide 3-Methylthio-1-propene | |
| Identifiers | |
| 10152-76-8 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 21159856 |
| ECHA InfoCard | 100.030.371 |
| EC Number | 233-422-0 |
| MeSH | allyl+methyl+sulfide |
| PubChem | 66282 |
| RTECS number | UD1015000 |
| UNII | V7QI1R316C |
| UN number | 1993 |
| |
| |
| Properties | |
| C4H8S | |
| Molar mass | 88.17 g·mol−1 |
| Odor | Garlic |
| Density | 0.803 g cm−3 |
| Boiling point | 92 °C; 197 °F; 365 K |
| Hazards | |
| GHS pictograms |  |
| GHS signal word | DANGER |
| H225 | |
| P210 | |
| EU classification (DSD) |
|
| R-phrases | R11 |
| S-phrases | S16, S29, S33 |
| Flash point | 18.0 °C (64.4 °F; 291.1 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Allyl methyl sulfide (AMS) is an organosulfur compound with the chemical formula CH2=CHCH2SCH3. The molecule features two functional groups, an allyl (CH2=CHCH2) and a sulfide. It is a colourless liquid with a strong odor characteristic of alkyl sulfides. It is a metabolite of garlic, and "garlic breath" is attributed to its presence.[1]
It is prepared by the reaction of allyl chloride with sodium hydroxide and methanethiol.
- CH2=CHCH2Cl + NaOH(aq) + CH3SH → CH2=CHCH2SCH3 + NaCl + H2O
References
This article is issued from Wikipedia - version of the 8/8/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.