Acetosyringone
 | |
| Names | |
|---|---|
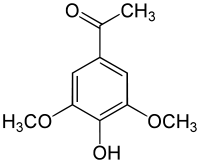
| IUPAC name
4'-Hydroxy-3',5'-dimethoxyacetophenone | |
| Other names
Acetosyringenin | |
| Identifiers | |
| 2478-38-8 | |
| 3D model (Jmol) | Interactive image Interactive image |
| ChEBI | CHEBI:2404 |
| ChEMBL | ChEMBL224146 |
| ChemSpider | 16280 |
| ECHA InfoCard | 100.017.828 |
| PubChem | 1 |
| |
| |
| Properties | |
| C10H12O4 | |
| Molar mass | 196.20 g·mol−1 |
| Melting point | 125.5 °C (257.9 °F; 398.6 K) |
| Boiling point | 335 °C (635 °F; 608 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Acetosyringone is a phenolic natural product, and is a chemical compound related to acetophenone and 2,6-dimethoxyphenol. It was first described in relation to lignan/phenylpropanoid-type phytochemicals, with isolation from a variety of plant sources, in particular, in relation to wounding and other physiologic changes.
Historically, this substance has been best known for its involvement in plant-pathogen recognition,[1] especially its role as a signal attracting and transforming unique, oncogenic bacteria in genus Agrobacterium. The virA gene on the Ti plasmid of Agrobacterium tumefaciens and the Ri plasmid of Agrobacterium rhizogenes is used by these soil bacteria to infect plants, via its encoding for a receptor for acetosyringone and other phenolic phytochemicals exuded by plant wounds.[2] This compound also allows higher transformation efficiency in plants, in A. tumefaciens-mediated transformation procedures, and so is of importance in plant biotechnology.[3]
Acetosyringone can also be found in Posidonia oceanica[4] and a wide variety of other plants. It is secreted at wounded site of dicotyledons. This compound enhances the Agrobacterium-mediated gene transformation in dicot. Monocotyledons lack this wound response and it is considered as the limiting factor in Agrobacterium-mediated gene transformation in monocots.[5]
The compound is also produced by the male leaffooted bug (Leptoglossus phyllopus) and used in its communication system.[6][7][8]
In vitro studies show that acetosyringone increases mycorrhizae formation in the fungus Glomus intraradices.[9]
Total synthesis of this simple natural product performed by Crawford et al. in 1956,[10] but is of limited contemporary synthetic interest. A variety of acetosyringone analogs are available, including some which are covalent inactivators of cellular processes that involve acetosyringone.
Chemical characteristics
Acetosyringone does not dissolve well in water. Although it has a melting point of about 125 degree Celsius, it is not wise to autoclave acetosyringone along with the medium used for (for example) plant infiltration by microbes.
See also
References
- ↑ Baker C. Jacyn; Mock Norton M.; Whitaker Bruce D.; Roberts Daniel P.; Rice Clifford P.; Deahl Kenneth L.; Aver'Yanov Andrey A. (2005). "Involvement of acetosyringone in plant-pathogen recognition". Biochemical and Biophysical Research Communications. 328 (1): 130–136. doi:10.1016/j.bbrc.2004.12.153. PMID 15670760.
- ↑ Schrammeijer, B.; Beijersbergen, A; Idler, KB; Melchers, LS; Thompson, DV; Hooykaas, PJ (2000). "Sequence analysis of the vir-region from Agrobacterium tumefaciens octopine Ti plasmid pTi15955". Journal of Experimental Botany. 51 (347): 1167–1169. doi:10.1093/jexbot/51.347.1167. PMID 10948245.
- ↑ Sheikholeslam, Shahla N.; Weeks, Donald P. (1987). "Acetosyringone promotes high efficiency transformation of Arabidopsis thaliana explants by Agrobacterium tumefaciens". Plant Molecular Biology. 8 (4): 291–298. doi:10.1007/BF00021308.
- ↑ Agostini, Sylvia; Desjobert, Jean-Marie; Pergent, Gérard (1998). "Distribution of phenolic compounds in the seagrass Posidonia oceanica". Phytochemistry. 48 (4): 611–617. doi:10.1016/S0031-9422(97)01118-7.
- ↑ Naveed Iqbal Raja; Asghari Bano; Hamid Rashid; Zubeda Chaudry; Noshin Ilyas (2010). "Improving Agrobacterium-mediated Transformation Protocol For Integration Of XA21 Gene In Wheat (Triticum aestivum L.)" (PDF). Pak. J. Bot. 42 (5): 3613–3631.
- ↑ Acetosyringone on www.pherobase.com, the pheromones data base
- ↑ Aldrich, J.R.; Blum, M.S.; Duffey, S.S.; Fales, H.M. (1976). "Male specific natural products in the bug, Leptoglossus phyllopus: Chemistry and possible function". Journal of Insect Physiology. 22 (9): 1201–1206. doi:10.1016/0022-1910(76)90094-9.
- ↑ Aldrich, J. R.; Blum, M. S.; Fales, H. M. (1979). "Species-specific natural products of adult male leaf-footed bugs (Hemiptera: Heteroptera)". Journal of Chemical Ecology. 5: 53–62. doi:10.1007/BF00987687.
- ↑ Estela Flores-Gómez; Lidia Gómez-Silva; Roberto Ruiz-Medrano; Beatriz Xoconostle-Cázares (2008). "Role of acetosyringone in the accumulation of a set of RNAs in the arbuscular mycorrhiza fungus Glomus intraradices". International Microbiology. 11 (4): 275–282. doi:10.2436/20.1501.01.72. PMID 19204900.
- ↑ L. W. Crawford; E. O. Eaton; J. M. Pepper (1956). "An Improved Synthesis of Acetosyringone". Canadian Journal of Chemistry. 34 (11): 1562–1566. doi:10.1139/v56-204.