4-HO-MET
 | |
 | |
| Names | |
|---|---|
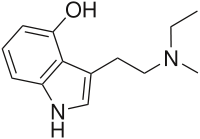
| IUPAC name
3-{2-[Ethyl(methyl)amino]ethyl}-1H-indol-4-ol | |
| Other names
4-hydroxy-N-methyl-N-ethyltryptamine; metocin; 3-{2-[ethyl(methyl)amino]ethyl}-1H-indol-4-ol | |
| Identifiers | |
| 77872-41-4 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 10513072 |
| PubChem | 21786582 |
| UNII | 6RN01B78NY |
| |
| |
| Properties | |
| C13H18N2O | |
| Molar mass | 218.30 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
4-HO-MET (4-hydroxy-N-methyl-N-ethyl tryptamine, or metocin, methylcybin, Colour), is a lesser-known psychedelic drug. It is a structural− and functional analog of psilocin as well as the 4-hydroxyl analog of MET. 4-HO-MET was first synthesized by Alexander Shulgin. In his book TiHKAL (Tryptamines I Have Known and Loved), the dosage is listed as 10-20 mg.[1] 4-HO-MET produces psilocin-like distortion of color, sound, and form. Very little data exists about the pharmacological properties, metabolism, and toxicity of 4-HO-MET. There have been no reports of deaths from 4-HO-MET, even though people have reported taking doses up to 150 mg,[2] more than an order of magnitude above the effective dose.[3]
Effects
Users report similar effects to psilocin, including mydriasis, closed and open eye visuals, euphoria, time dilation and general change in thought processes. These effects occur in a wavelike pattern such as that of psilocybin with near-normal perception and high effect varying rapidly. The effects last for about 4–6 hours.[4]
Drug prohibition laws
Sweden
Sveriges riksdag added 4-HO-MET to schedule I ("substances, plant materials and fungi which normally do not have medical use") as narcotics in Sweden as of May 1, 2012, published by Medical Products Agency in their regulation LVFS 2012:6 listed as 4-HO-MET 3-[2-[etyl(metyl)amino]etyl]-1H-indol-4-ol.[5]
United Kingdom
4-HO-MET is a class A drug in the UK, as a result of the tryptamine catch-all clause.
See also
References
- ↑ Shulgin, Alexander; Ann Shulgin (September 1997). TiHKAL: The Continuation. Berkeley, California: Transform Press. ISBN 0-9630096-9-9. OCLC 38503252.
- ↑ "Erowid Experience Vaults / Cosmic Mind Orgy". Erowid. 2008-07-22. Retrieved 2014-02-25.
- ↑ http://www.erowid.org/experiences/exp.cgi?S1=436&S2=-1&C1=-1&Str=
- ↑ "4-HO-MET Effects". Erowid. Retrieved 2014-02-25.
- ↑ "Föreskrifter om ändring i Läkemedelsverkets föreskrifter (LVFS 2011:10) om förteckningar över narkotika;" (PDF) (in Swedish). Elanders Sverige AB. 2012-04-20. Retrieved 2014-02-25.