1,8-Diazabicyclo(5.4.0)undec-7-ene
 | |
 | |
| Names | |
|---|---|
| IUPAC name
2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepine | |
| Other names
DBU,Diazabicycloundecene | |
| Identifiers | |
| 6674-22-2 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 73246 |
| ECHA InfoCard | 100.027.013 |
| |
| |
| Properties | |
| C9H16N2 | |
| Molar mass | 152.24 g/mol |
| Appearance | Colorless liquid |
| Density | 1.018 g/mL liquid |
| Melting point | −70 °C (−94 °F; 203 K) |
| Boiling point | 80 to 83 °C (176 to 181 °F; 353 to 356 K) (0.6 mmHg); 261 °C (1 atm) |
| Acidity (pKa) | 13.5±1.5[1] (pKa of conjugate acid in water); 24.34[2] (pKa of conjugate acid in acetonitrile) |
| Hazards | |
| R-phrases | R22 R34 |
| S-phrases | S24 S25 |
| Flash point | 119.9 °C (247.8 °F; 393.0 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
1,8-Diazabicyclo[5.4.0]undec-7-ene, or more commonly DBU, is a chemical compound and belongs to the class of amidine compounds. It is used in organic synthesis as a catalyst, a complexing ligand, and a non-nucleophilic base.
Occurrence
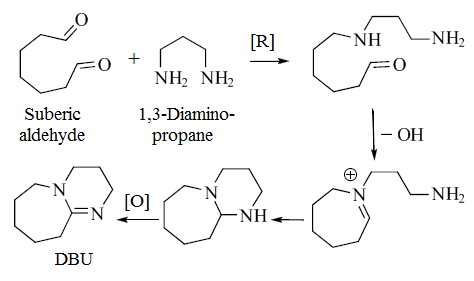
Although DBU is typically produced synthetically, it is also an alkaloid isolated from the sponge Niphates digitalis.[3] The biosynthesis of DBU has been proposed to begin with suberic aldehyde and diaminopropane.[3]
Uses
As a reagent in organic chemistry, DBU is used as a catalyst, a complexing ligand, and a non-nucleophilic base. It is also used as a curing agent for epoxy. It is used in fullerene purification with trimethylbenzene (it reacts with C70 and higher fullerenes, but not to C60 fullerenes); and it is also used as a catalyst in polyurethane production. It has a strong catalyst effect for the reactions of alicyclic and aliphatic isocyanates. It also exhibited its dual character (base and nucleophile) in the synthesis of aryl- & styryl- terminal acetylenes.
See also
References
- ↑ Kaupmees, K.; Trummal, A.; Leito, I. (2014). "Basicities of Strong Bases in Water: A Computational Study". Croat. Chem. Acta. 87: 385–395. doi:10.5562/cca2472.
- ↑ Kaljurand, I.; Kütt, A.; Sooväli, L.; Rodima, T.; Mäemets, V.; Leito, I.; Koppel, I. A. (2005). "Extension of the Self-Consistent Spectrophotometric Basicity Scale in Acetonitrile to a Full Span of 28 pKa Units: Unification of Different Basicity Scales". J. Org. Chem. 70: 1019–1028. doi:10.1021/jo048252w. PMID 15675863.
- 1 2 Regalado, E.L. et al., Nat. Prod. Commun., 2010, 5, 1187- 1190